Our websites may use cookies to personalize and enhance your experience. By continuing without changing your cookie settings, you agree to this collection. For more information, please see our University Websites Privacy Notice .

UConn Today
- School and College News
- Arts & Culture
- Community Impact
- Entrepreneurship
- Health & Well-Being
- Research & Discovery
- UConn Health
- University Life
- UConn Voices
- University News
September 7, 2021 | Combined Reports - UConn Communications
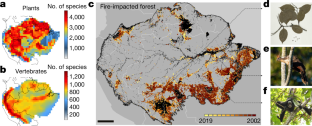
Study Shows the Impacts of Deforestation and Forest Burning on Biodiversity in the Amazon
Since 2001, between 40,000 and 73,400 square miles of Amazon rainforest have been impacted by fires

Ring of fire: Smoke rises through the understory of a forest in the Amazon region. Plants and animals in the Amazonian rainforest evolved largely without fire, so they lack the adaptations necessary to cope with it. (Credit: Paulo Brando)
A new study, co-authored by a team of researchers including UConn Ecology and Evolutionary Biology researcher Cory Merow provides the first quantitative assessment of how environmental policies on deforestation, along with forest fires and drought, have impacted the diversity of plants and animals in the Amazon. The findings were published in the Sept. 1 issue of Nature .
Researchers used records of more than 14,500 plant and vertebrate species to create biodiversity maps of the Amazon region. Overlaying the maps with historical and current observations of forest fires and deforestation over the last two decades allowed the team to quantify the cumulative impacts on the region’s species.
They found that since 2001, between 40,000 and 73,400 square miles of Amazon rainforest have been impacted by fires, affecting 95% of all Amazonian species and as many as 85% of species that are listed as threatened in this region. While forest management policies enacted in Brazil during the mid-2000s slowed the rate of habitat destruction, relaxed enforcement of these policies coinciding with a change in government in 2019 has seemingly begun to reverse this trend, the authors write. With fires impacting 1,640 to 4,000 square miles of forest, 2019 stands out as one of the most extreme years for biodiversity impacts since 2009, when regulations limiting deforestation were enforced.
“Perhaps most compelling is the role that public pressure played in curbing forest loss in 2019,” Merow says. “When the Brazilian government stopped enforced forest regulations in 2019, each month between January and August 2019 was the worse month on record (e.g. comparing January 2019 to previous January’s) for forest loss in the 20-year history of available data. However, based on international pressure, forest regulation resumed in September 2019, and forest loss declined significantly for the rest of the year, resulting in 2019 looking like an average year compared to the 20-year history. This was big: active media coverage and public support for policy changes were effective at curbing biodiversity loss on a very rapid time scale.”
The findings are especially critical in light of the fact that at no point in time did the Amazon get a break from those increasing impacts, which would have allowed for some recovery, says senior study author Brian Enquist, a professor in UArizona’s Department of Ecology and Evolutionary Biology .
“Even with policies in place, which you can think of as a brake slowing the rate of deforestation, it’s like a car that keeps moving forward, just at a slower speed,” Enquist says. “But in 2019, it’s like the foot was let off the brake, causing it to accelerate again.”
Known mostly for its dense rainforests, the Amazon basin supports around 40% of the world’s remaining tropical forests. It is of global importance as a provider of ecosystem services such as scrubbing and storing carbon from the atmosphere, and it plays a vital role in regulating Earth’s climate. The area also is an enormous reservoir of the planet’s biodiversity, providing habitats for one out of every 10 of the planet’s known species. It has been estimated that in the Amazon, 1,000 tree species can populate an area smaller than a half square mile.
“Fire is not a part of the natural cycle in the rainforest,” says study co-author Crystal N. H. McMichael at the University of Amsterdam. “Native species lack the adaptations that would allow them to cope with it, unlike the forest communities in temperate areas. Repeated burning can cause massive changes in species composition and likely devastating consequences for the entire ecosystem.”
Since the 1960s, the Amazon has lost about 20% of its forest cover to deforestation and fires. While fires and deforestation often go hand in hand, that has not always been the case, Enquist says. As climate change brings more frequent and more severe drought conditions to the region, and fire is often used to clear large areas of rainforest for the agricultural industry, deforestation has spillover effects by increasing the chances of wildfires. Forest loss is predicted reach 21 to 40% by 2050, and such habitat loss will have large impacts on the region’s biodiversity, according to the authors.
“Since the majority of fires in the Amazon are intentionally set by people, preventing them is largely within our control,” says study co-author Patrick Roehrdanz, senior manager of climate change and biodiversity at Conservation International. “One way is to recommit to strong antideforestation policies in Brazil, combined with incentives for a forest economy, and replicate them in other Amazonian countries.”
Policies to protect Amazonian biodiversity should include the formal recognition of Indigenous lands, which encompass more than one-third of the Amazon region, the authors write, pointing to previous research showing that lands owned, used or occupied by Indigenous peoples have less species decline, less pollution and better-managed natural resources.
The authors say their study underscores the dangers of continuing lax policy enforcement. As fires encroach on the heart of the Amazon basin, where biodiversity is greatest, their impacts will have more dire effects, even if the rate of forest burning remains unchanged.
The research was made possible by strategic investment funds allocated by the Arizona Institutes for Resilience at UArizona and the university’s Bridging Biodiversity and Conservation Science group. Additional support came from the National Science Foundation’s Harnessing the Data Revolution program . Data and computation were provided through the Botanical Information and Ecology Network , which is supported by CyVerse , the NSF’s data management platform led by UArizona.
Recent Articles

June 10, 2024
Being an Active Ally: Not Just a June Activity
Read the article

Iranian Pair Designing App to Help International Students Navigate New Campus, New Life

June 7, 2024
Revolutionizing Nerve Damage Treatment
| GEOG 30N Environment and Society in a Changing World |

- ORIENTATION
- ASSIGNMENTS
- Program Home Page
- LIBRARY RESOURCES
- Getting Help
- Engaging Course Concepts
Case Study: The Amazon Rainforest
The Amazon in context
Tropical rainforests are often considered to be the “cradles of biodiversity.” Though they cover only about 6% of the Earth’s land surface, they are home to over 50% of global biodiversity. Rainforests also take in massive amounts of carbon dioxide and release oxygen through photosynthesis, which has also given them the nickname “lungs of the planet.” They also store very large amounts of carbon, and so cutting and burning their biomass contributes to global climate change. Many modern medicines are derived from rainforest plants, and several very important food crops originated in the rainforest, including bananas, mangos, chocolate, coffee, and sugar cane.

In order to qualify as a tropical rainforest, an area must receive over 250 centimeters of rainfall each year and have an average temperature above 24 degrees centigrade, as well as never experience frosts. The Amazon rainforest in South America is the largest in the world. The second largest is the Congo in central Africa, and other important rainforests can be found in Central America, the Caribbean, and Southeast Asia. Brazil contains about 40% of the world’s remaining tropical rainforest. Its rainforest covers an area of land about 2/3 the size of the continental United States.
There are countless reasons, both anthropocentric and ecocentric, to value rainforests. But they are one of the most threatened types of ecosystems in the world today. It’s somewhat difficult to estimate how quickly rainforests are being cut down, but estimates range from between 50,000 and 170,000 square kilometers per year. Even the most conservative estimates project that if we keep cutting down rainforests as we are today, within about 100 years there will be none left.
How does a rainforest work?
Rainforests are incredibly complex ecosystems, but understanding a few basics about their ecology will help us understand why clear-cutting and fragmentation are such destructive activities for rainforest biodiversity.

High biodiversity in tropical rainforests means that the interrelationships between organisms are very complex. A single tree may house more than 40 different ant species, each of which has a different ecological function and may alter the habitat in distinct and important ways. Ecologists debate about whether systems that have high biodiversity are stable and resilient, like a spider web composed of many strong individual strands, or fragile, like a house of cards. Both metaphors are likely appropriate in some cases. One thing we can be certain of is that it is very difficult in a rainforest system, as in most other ecosystems, to affect just one type of organism. Also, clear cutting one small area may damage hundreds or thousands of established species interactions that reach beyond the cleared area.
Pollination is a challenge for rainforest trees because there are so many different species, unlike forests in the temperate regions that are often dominated by less than a dozen tree species. One solution is for individual trees to grow close together, making pollination simpler, but this can make that species vulnerable to extinction if the one area where it lives is clear cut. Another strategy is to develop a mutualistic relationship with a long-distance pollinator, like a specific bee or hummingbird species. These pollinators develop mental maps of where each tree of a particular species is located and then travel between them on a sort of “trap-line” that allows trees to pollinate each other. One problem is that if a forest is fragmented then these trap-line connections can be disrupted, and so trees can fail to be pollinated and reproduce even if they haven’t been cut.
The quality of rainforest soils is perhaps the most surprising aspect of their ecology. We might expect a lush rainforest to grow from incredibly rich, fertile soils, but actually, the opposite is true. While some rainforest soils that are derived from volcanic ash or from river deposits can be quite fertile, generally rainforest soils are very poor in nutrients and organic matter. Rainforests hold most of their nutrients in their live vegetation, not in the soil. Their soils do not maintain nutrients very well either, which means that existing nutrients quickly “leech” out, being carried away by water as it percolates through the soil. Also, soils in rainforests tend to be acidic, which means that it’s difficult for plants to access even the few existing nutrients. The section on slash and burn agriculture in the previous module describes some of the challenges that farmers face when they attempt to grow crops on tropical rainforest soils, but perhaps the most important lesson is that once a rainforest is cut down and cleared away, very little fertility is left to help a forest regrow.
What is driving deforestation in the Amazon?
Many factors contribute to tropical deforestation, but consider this typical set of circumstances and processes that result in rapid and unsustainable rates of deforestation. This story fits well with the historical experience of Brazil and other countries with territory in the Amazon Basin.
Population growth and poverty encourage poor farmers to clear new areas of rainforest, and their efforts are further exacerbated by government policies that permit landless peasants to establish legal title to land that they have cleared.
At the same time, international lending institutions like the World Bank provide money to the national government for large-scale projects like mining, construction of dams, new roads, and other infrastructure that directly reduces the forest or makes it easier for farmers to access new areas to clear.
The activities most often encouraging new road development are timber harvesting and mining. Loggers cut out the best timber for domestic use or export, and in the process knock over many other less valuable trees. Those trees are eventually cleared and used for wood pulp, or burned, and the area is converted into cattle pastures. After a few years, the vegetation is sufficiently degraded to make it not profitable to raise cattle, and the land is sold to poor farmers seeking out a subsistence living.
Regardless of how poor farmers get their land, they often are only able to gain a few years of decent crop yields before the poor quality of the soil overwhelms their efforts, and then they are forced to move on to another plot of land. Small-scale farmers also hunt for meat in the remaining fragmented forest areas, which reduces the biodiversity in those areas as well.
Another important factor not mentioned in the scenario above is the clearing of rainforest for industrial agriculture plantations of bananas, pineapples, and sugar cane. These crops are primarily grown for export, and so an additional driver to consider is consumer demand for these crops in countries like the United States.
These cycles of land use, which are driven by poverty and population growth as well as government policies, have led to the rapid loss of tropical rainforests. What is lost in many cases is not simply biodiversity, but also valuable renewable resources that could sustain many generations of humans to come. Efforts to protect rainforests and other areas of high biodiversity is the topic of the next section.
- Make a Donation
- Donate in Someone's Name
- Renew your Membership
- Join as a Partner in Conservation
- Respond to our TV Ad
- See Other Ways to Support
- What we care about
- Climate crisis
- Sustainability
- How we work
- Public policy
- Wildlife conservation
WWF works to sustain the natural world for the benefit of people and wildlife, collaborating with partners from local to global levels in nearly 100 countries.
- Take action
- Send ecards
- Educational resources
- Leave a legacy gift
- Travel with us
- Leadership team
- Board & Council
Stories & updates
World Wildlife Magazine
The Amazon in crisis: Forest loss threatens the region and the planet
We must act now to protect the amazon from catastrophic and irreversible damage.

- Date: November 08, 2022
The numbers are devastating: 17% of Amazon forests have been wholly lost, and an additional 17% are degraded. And data from the first half of 2022 show the loss continuing to grow.
The Amazon is in crisis as forests are threatened by deforestation, fires, and degradation; surface water has been lost; and rivers are increasingly disconnected and polluted. This immense pressure—if not slowed or stopped—will irreversibly damage the Amazon and the overall planet in the very near future.
With the launch of its Living Amazon Report 2022 , WWF synthesizes the latest information on the region, its critical global role, the threats it faces, and solutions that require an unprecedented global commitment to stop the Amazon’s destruction.
What’s at stake
As the world’s largest tropical forest and river system, the Amazon is an unparalleled and complex biome:
People: 47 million people live in the Amazon region and depend upon it for their livelihoods. This includes 2.2 million Indigenous peoples from more than 500 different groups.
Biodiversity and wildlife: The Amazon is home to a stunning array of the world’s species: 9% of mammals; 14% of birds; 8% of amphibians; 13% of freshwater fish species; and 22% of vascular plant species. Many of these species are found nowhere else in the world, and scientists estimate there are places in the Amazon where up to 90% of the species are yet to be discovered.

Lucinézio Cerdeira de Melo, a teacher; his daughter Louise Sophia; and wife Luciane Melo; are part of the Suruacá village community.

While there are many free-flowing rivers in the Amazon basin, hundreds of dams within the river network threaten connectivity and water quality.
Forests: Without its forests, the Amazon region would lose its biodiversity, release massive amounts of carbon, suffer soil erosion, and face hydrological and climatic devastation. Without its ecosystem services, local communities and people around the world would face a loss of livelihoods, well-being, and ecological stability.
Climate: The Amazon is a major carbon sink that regulates and helps stabilize the planet’s climate. Any loss or degradation of its forests means an increase in carbon emissions. Today, land conversion and fires in the region are already releasing some of that carbon into the atmosphere at record highs.
Food: The Amazon’s “flying rivers” transport moisture outside of the basin to the southern part of the continent, providing the necessary conditions for agriculture in Argentina, Paraguay, Uruguay, and southern Brazil. The health and vitality of the Amazon River basin are also fundamental locally for the millions of people who rely on its waterways for fish.
Freshwater: The Amazon is the largest free-flowing river in the world. Home to 20% of the freshwater discharged into the Earth's oceans, the Amazon must stay free flowing and healthy. Its connectivity and water quality impact not only the river basin but also human health, food security, livelihoods, and the mangroves and surrounding wetlands the river passes on its way to the Atlantic Ocean.
Ancestral knowledge: People have lived in the Amazon for more than 12,000 years, making the region a rich repository of human history and ancestral culture.

The Amazon rivers and forests are home to nearly 10 percent of the world's biodiversity, including squirrel monkeys.

Acai berries are one of the Amazon's many resources produced for human consumption.
The health of the Amazon has both local and far-reaching impacts. Losing the Amazon would drastically change the climate of South America, worsening food security, intensifying the climate crisis, and ultimately affecting the entire planet. The global climate emergency would accelerate, as keeping planetary warming below 1.5 degrees Celsius would be impossible.
Reversing the trend
As a first step, we must recognize the interdependence between ourselves and the Amazon. Our future hinges on its survival, and the Amazon depends on us and the choices we make today.
WWF’s Living Amazon report proposes strategies to reverse current losses and ways for governments, the private sector, and everyday citizens to take urgent action for the Amazon and its conservation. This includes the 80x25 initiative, which aims to conserve 80% of the Amazon by 2025. The plan was presented by the Coordinator of the Indigenous Organizations of the Amazon Basin, adopted as an IUCN motion in 2021, and supported by WWF, but will require further commitment at all levels to succeed.
Amazonian countries must agree to and prioritize the protection and sustainable management of the Amazon and its cross-border and interconnected systems. Indigenous peoples must be included in decision-making for the region.
Corporations that profit from the Amazon’s natural resources should examine their supply chains and ensure sustainable practices are enforced. Consumers can change their consumption patterns and refuse to purchase products that drive deforestation and land conversion in the Amazon.
Together, we can turn the tide on Amazon loss and shift toward social equity, inclusive economic development, and global responsibility.
Read the full Living Amazon Report 2022

Take Action
Join us to make change. Speak up for species and places through WWF's Action Center.

Give to WWF
Make a gift to WWF's global conservation efforts and choose from symbolic adoptions, apparel, and more!

- Latest stories
- Adopt an animal
- Give in someone's name
- Gift and estate planning
- Get involved
- Help center
- WWF country offices
- Ethics reporting
World Wildlife Fund
1250 24th Street, N.W. Washington, DC 20037
- Connect with us
World Wildlife Fund Inc. is a nonprofit, tax-exempt charitable organization (tax ID number 52-1693387) under Section 501(c)(3) of the Internal Revenue Code. Donations are tax-deductible as allowed by law.
- Privacy Policy/Your Privacy Rights
- State Disclosures
© 2024 World Wildlife Fund. WWF ® and ©1986 Panda Symbol are owned by WWF. All rights reserved.
Accessibility Settings
Color options, reading tools.

An aerial view showing how agricultural land is encroaching on the forest in Brazil, with illegal burning in the background. Image: Courtesy of Fernando Martinho
Investigating ‘Cattle Laundering’ and Deforestation in the Amazon: Interview with the 2024 Goldman Prize Winner
Read this article in.
- Português ( Portuguese (Brazil) )
Related Resources

The Investigative Agenda for Climate Change

Remote Sensing and Data Tools for Environmental Investigations

GIJC23 – How to Use Satellite Images for Investigation

Climate Change: Investigating the Story of the Century
For years Brazil’s rainforests have been under attack, with satellite imagery showing the borders of the Amazon jungle slowly, but steadily, receding.
In 2022, deforestation in the country was the highest of anywhere in the world , according to data on tropical primary forest loss from Global Forest Watch.
There are many motivations for transforming forests into fields, ranging from mining to the construction of hydroelectric plants, but the main driver is converting forest to pasture for raising cattle .
Brazil also tops another list, as the world’s largest exporter of beef, exporting to more than 150 countries according to government figures. Last year alone, around 2.5 million tons of fresh and processed beef were sent abroad.
But although the link between deforestation and the sale of beef is widely understood, especially among Brazilians, finding documentary evidence in the paperwork — and tracking the supply chain from point of origin to consumer — is not as simple as it seems.
Often, animals pass through several farms, in different states of the country, before being slaughtered. It’s a process called “cattle laundering” and it makes tracking the livestock practically impossible.
In a multi-year investigation , journalists from Repórter Brasil — a GIJN member since 2018 — worked with researchers from different countries to document the supply chain back from the supermarket to the fields where the cattle are raised, and used data on environmental fines and satellite images on farm locations to track the movement of cattle and beef products to deforestation in Brazil.
That eventually helped the outlet reveal links between supermarket chains in Europe and the United States with Brazilian slaughterhouses supplied by livestock farmers charged with illegal deforestation in different biomes, such as the Amazon, Cerrado, and Pantanal.
After the story was published in 2021, six large retail groups from Belgium, France, the Netherlands, and the United Kingdom suspended the sale of products from the Brazilian meat companies in question.

Marcel Gomes, executive secretary of Repórter Brasil. Photo: Courtesy of Goldman Environmental Prize
“About 65% of the deforestation in the Amazon region is caused by cattle ranching,” explained Marcel Gomes, the executive secretary of Repórter Brasil, who led the project. “It’s our duty in journalism to expose these problems, to hold the authorities and companies accountable.”
The exposé earned Gomes the 2024 Goldman Environmental Prize . Known as the “Nobel of Environmentalism,” Gomes is the first Brazilian journalist to win the award, which is usually given to activists and environmental defenders.
Repórter Brasil’s dogged investigations into topics including deforestation, the environment, and human rights have also earned the outlet a number of journalism awards and recognitions, including a 2023 Gabriel García Márquez Prize and a finalist nomination for the 2023 Javier Valdez Prize .
We spoke with Gomes to understand how the project was carried out, the tips and tools they used to follow the cattle trail, and the risks of reporting on this topic where huge profits — and reputations — are at stake.

Marcel Gomes and colleagues Natália Suzuki (left) and Tatiana Chang Waldman at the Reporter Brasil office. Photo: Courtesy of Goldman Environmental Prize
GIJN: Your investigation dug into meat supply chains and deforestation. In general, how important is the issue of livestock farming in environmental coverage?
Marcel Gomes: It is very important because it is a sector that has a lot of impact. The sector is responsible for deforestation in the Amazon, cases of work similar to slavery, and is also relevant in the country’s economy, both for meat, as we are the largest exporter of meat, and also for the export of biodiesel, which is made with animal fat, and leather, from clothing to automobiles.
Our innovation was to address the issue by considering the entire production chain. It is a very long chain: a cow is born on a farm, from this farm it goes to another farm to be raised, then to another farm where it is fattened, and it often passes through others until it reaches the slaughterhouse. This means that something very important is lost when we talk about the production chain, which is traceability. You don’t know the path this animal has taken throughout its life. At the same time, you have a series of social and environmental impacts linked to this chain. Without traceability you hardly have any influence to improve the situation.
This investigation won awards because we went a step further: we involved the supermarkets. We hired researchers in several countries, including here in Brazil, who went to shops to photograph the barcodes on the packaging. We fed that information into a large spreadsheet that took us to the slaughterhouses where these Brazilian meats spread around the world had originated.
GIJN: Your team created software to track the ‘meat route.’ How did you go about that?
MG: We have had a Public Information Law here in Brazil since 2011. This law requires public bodies to digitize their information. This means that you have a huge volume of information, but this data is super disorganized. Each [piece of data] is on a different server, in a different language, organized in different formats and even in different formats: one is in PDF, another in Word, another in Excel.
Repórter Brasil has been creating databases from the beginning. In 2013, we launched a database on the textile chain in Brazil, Moda Livre, which showed the companies’ links with slave labor and other labor relations. This is available and updated periodically. The secret is to have the ability to organize the data in a way that allows you to perform faster searches and identify headlines and stories. Our partners have access to this database so they can access and search for information. Our database on cattle is updated once a year for those who support Repórter Brasil.

Aerial view of cattle confinement used by farms in the Amazon. Photo: Courtesy of Fernando Martinho
GIJN: The cattle deforestation report had an economic impact as companies stopped buying meat from Brazil. Did you face any retaliation?
MG: The methodology that Repórter Brasil used was to listen to everyone involved. We had a very frank dialogue, both with the meatpacking sector and with supermarkets… [where we presented] data, proof, and evidence.
We did not suffer any type of pressure or retaliation. The biggest risk in this type of investigation is with livestock farmers… and we have had some lawsuits filed by livestock farmers from other reports.
This is a tip I always give: it is essential to have a good team of legal professionals. It is very difficult to do this type of work independently, without the support of legal professionals, because companies in the private sector, or even politicians, sue you. Some of our publications are evaluated by lawyers beforehand, as was the case with this material. That’s why we have a lot of security when we publish material like this. We believe in justice.
GIJN: Do you think that environmental journalism brings a double risk: that of legal harassment and danger in the field?
MG: If you do a risk analysis, there are many things involved with a field trip, with safety in relation to criminals or accidents, right? And in the period after you have the risk of legal harassment, lawsuits, threats. We have a security policy, a risk checklist. We have a security policy that is very time-consuming, but it is essential to avoid tragedies. Recently Repórter Brasil reporters were detained by police thugs who had ties to the militia. We had to send a lawyer to the scene once to free one of our reporters. After the death of Indigenous researcher Bruno Pereira and journalist Dom Philips , we didn’t travel for a long time. Then we strengthened our security policy. We introduced even more criteria that ended up making travel even more expensive. But it’s necessary.
Watch Marcel Gomes discuss his investigation into how cattle ranching fuels illicit deforestation in the Amazon, on YouTube below.
GIJN: How would you describe Repórter Brasil?
MG: Repórter Brasil is an organization that was created when we were still students at the journalism faculty at the University of São Paulo. We started to discuss the possibility of creating a vehicle that could work on social and environmental topics, topics we thought that the mainstream press did not adequately cover. Repórter Brasil was founded in 2001 and is a unique organization because it does journalism and at the same time is an NGO. We have a research program, an education program, and a political advocacy program. One of the strategies we have is to try to enhance journalistic content so that it really causes impact and social transformation. So, when we carry out an investigation, this material is often used not only by other areas of Repórter Brasil but by our partners. We are producing journalistic work, but at the same time we are fighting for that content to change something in a real way. RB is seen as an outlet, we don’t have copyright, and supply material to UOL, Folha de S. Paulo, and the Guardian. I think it’s a very interesting organizational model.
GIJN: How does this model differ from that of traditional media organizations, and how does that help you?
MG: For us, the ethical issue is fundamental. Although Repórter Brasil is part of a coalition of non-governmental organizations and participates in conversations, our journalism program is independent. Our content is ours, produced by us, and edited by us. We insist on maintaining this way, which is to protect journalistic work so that it is independent. This is why Repórter Brasil has an excellent reputation and is able to supply material to traditional press organizations. The agendas are defined based on the coverage we provide, the perspective we have on the problems: thinking about human rights, labor, and environmental rights.

Related Stories

Abraji: 20 Years Fighting for Investigative Journalists and Journalism in Brazil

Thriving on Change: El Surtidor’s Groundbreaking Multi-Platform, Visual Journalism

Peru’s Investigative Journalists Carry On Amidst Disinformation, Attacks, and Political Crisis

Investigating Environmental Crimes: Tips From Reporters Covering the Amazon
This work is licensed under a Creative Commons Attribution-NoDerivatives 4.0 International License
Republish our articles for free, online or in print, under a Creative Commons license.
Read other stories tagged with:
Republish this article, news & analysis.
Since its inception 20 years ago, the Brazilian Association of Investigative Journalism (Abraji) has remained faithful to its founding principles: professional training, defense of freedom of expression, and the right of access to public information.
Case Studies Member Profiles
GIJN member El Surtidor is a Paraguayan news organization created in 2015 that prioritizes innovation and multi-platform, visual journalism.
News & Analysis Safety & Security
Amidst disinformation and numerous attacks on press freedom, investigative reporting has all but disappeared from Peru’s major news outlets, leaving a handful of small nonprofit digital outlets to carry the mantle of accountability reporting.
GIJC23 Reporting Tools & Tips
Two reporters whose investigative work has exposed systemic land grabbing and illegal mining in the Amazon share their tips.
- Español (Spanish)
- Français (French)
- Bahasa Indonesia (Indonesian)
- Brasil (Portuguese)
- हिंदी (Hindi)
Mongabay Series: Amazon Conservation
Deforestation, climate crisis could crash Amazon tree diversity: study
- New research finds that when climate change and deforestation impacts are taken together, up to 58 percent of Amazon tree species richness could be lost by 2050, of which 49 percent would have some degree of risk for extinction.
- Under the deforestation/climate change scenario, half the Amazon (the north, central and west) could be reduced to 53 percent of the original forest. The other half (the east, south and southeast, where agribusiness occurs), could become extremely fragmented, with only 30 percent of forest remaining.
- Studies rarely take both climate change and deforestation into account. But the new study’s results bolster the findings of other scientists who have modeled results showing that when the Amazon is 20-25 percent deforested, it could cross a rainforest to savanna conversion tipping point, a disaster for biodiversity.
- Scientists warn that Jair Bolsonaro’s anti-environmental policies could result in a worst-case scenario, with severe damage to the Amazon rainforest and to its ecological services, including the loss of the sequestration of vast amounts of stored carbon, leading to a regional and global intensification of climate change.

The combined impact of ongoing deforestation and escalating climate change on the Amazon rainforest could radically transform its configuration by 2050, with the biome divided into two distinct blocks — one occupied by still significant but very seriously diminished rainforest, the other dominated by agribusiness and scattered forest inside conserved areas.
That shift, were it to occur, could result in a decline of up to 58 percent of Amazon tree species richness, of which 49 percent would have some degree of risk for extinction (with tree species becoming vulnerable, endangered or critically endangered), according to a new study published in Nature Climate Change .
The authors of the paper — four researchers from Brazil and the Netherlands — determined that both deforestation and climate change had to be examined together, and not separately as is typically done, in order to determine a realistic future scenario.
To the researchers’ surprise, when the effects were combined in their models, the tree species loss numbers were very high.
Under the deforestation/climate change scenario, one half of the Amazon (the northern, central and western portions) would be reduced to 53 percent of the original forest, although still with continuous areas. The other half (the eastern, southern and southeastern areas, where agribusiness activities currently take place), would become extremely fragmented, with only 30 percent of forest remaining; remnant vegetation would be found primarily in protected areas and indigenous reserves.

Two scenarios for 2050 were highlighted by the study, a more optimistic one — in which Paris Climate Agreement carbon targets are achieved and global temperature warms by less than 2 degrees Celsius (3.6 degrees Fahrenheit) — and a more pessimistic one based on recent rising trends in deforestation and CO 2 emissions.
In the more pessimistic scenario, Amazonian tree species would lose up to 65 percent of their spatial distribution area (where they live and reproduce), and up to 58 percent of their diversity; 49 percent would be threatened with extinction, of which 22 percent would be critically endangered, according to IUCN’s threatened extinction criteria.
Even the more optimistic scenario “does not indicate a promising future,” Vitor Gomes, an environmental scientist at the Federal University of Pará and the lead study author told Mongabay. That scenario predicts a loss of tree species richness of up to 43 percent, and a decline in tree species distribution area of up to 53 percent; under that scenario, 48 percent of tree species would be threatened, of which 11 percent would be critically endangered.
“The study should be seen as a big warning,” said Ima Vieira, a researcher at the Museu Paraense Emílio Goeldi and a study co-author. “It shows that if deforestation is currently the biggest cause of habitat loss in the Amazon, over the next thirty years it will probably be surpassed by climate change, which operates throughout the whole biome and can alone reduce species diversity by up to 37 percent.”
“It was not so clear to us how much the climate could affect the forest in the future,” Gomes added. “Deforestation is no longer the only major threat to the Pan-Amazon,” a biome designation that includes portions of nine South American countries.
Excluding climate change impacts, deforestation alone could cause Amazon species diversity losses of 19 percent (in the best scenario) or 36 percent (in the worst), while climate change could cause reductions of 31 percent to 37 percent, respectively.

No time for migration
The researchers analyzed the current distribution area of 6,394 tree species with available data among the 10,071 known Amazon tree species. They then compared that present data with both historical data (1950-2000) and projected deforestation data (up to 2050), along with current and future climate scenarios as determined by the United Nations IPCC (UN Intergovernmental Panel on Climate Change).
From this total species data, they eliminated rare species with insufficient records available to produce distribution models, and species without statistically significant models, leaving a total of 4,935 species.
Mapping the locations where the species can be found today is important to obtain temperature and precipitation limits in which they are apparently comfortable.
“The trees will find less favorable conditions [as climate change escalates] to keep existing and propagating,” explained Gomes. “Because they are static, they are slow to migrate to new areas through dispersers and pollinators [such as wind, water and animals].” The study gives as a reference the Holocene, a geological period that started about 11,600 years ago, when climate change caused Amazonian tree communities to expand their distribution southward.
“It took them 3,000 years to advance nearly 100 kilometers (62 miles). Man-induced climate change is happening now, and trees will be unable to move in 30 years or so more than 300 kilometers (186 miles), the distance to which the most suitable climates may be in relation with the current distribution areas [by 2050]. Not to mention that deforested areas [caused by agribusiness and other human development] make it even more difficult for trees to move forward once that barrier has been created,” Gomes said.
Tree species found in the lower half of the Amazon — such as the Protium altissimum (Aubl.) Marchand, the second most abundant in the biome, for example — may face a serious threat of extinction as the species suffers losses of up to 50 percent of its suitable distribution area. While Eperua falcata Aubl ., common in the Guiana Shield, could lose up to 63 percent of its suitable distribution area.

Agreement with previous studies
Carlos Nobre, a respected Brazilian climatologist and senior researcher at the University of São Paulo, along with Thomas Lovejoy, a U.S. ecologist at George Mason University, have estimated that deforestation and climate change combined could cause a large part of the Amazon biome to experience a rainforest to savanna conversion tipping point when around 20 to 25 percent of the biome is deforested.
Nobre commented on the new study: “The results are quite credible as projections of the synergistic effect of climate change, due to global warming, and deforestation, on the distribution of species in the Amazon. In our studies, we looked at the rainforest as a biome, not species by species, as in their study, and we analyzed the risks of change in the type of vegetation — that is, the forest being replaced by a savanna.”
However, “The results of these two kinds of analysis are similar: higher impacts in the south and east of the Amazon, while the forest would remain in the west,” Nobre said. “The study strongly reinforces our projections and recommendations for a development model with zero deforestation or, better still, the restoration of large deforested areas” in the Amazon.
Paulo Brando, an ecologist at the Woods Hole Research Center in Massachusetts, USA, noted that “the division of the Amazon in the middle is not necessarily a surprise. Previous studies, such as Duffy et al. 2015 , have shown that the Eastern Amazon could be[come] drier and hotter with climate change, while the west would be more rainy. That said, the results are of immense importance for the conservation of the rainforest.
“With the reduction of deforestation, Brazil could avoid the loss of occupation areas of hundreds of species. Without a global effort to stabilize the climate, however, some species would still be threatened. Thus, reducing deforestation would help not only to stabilize the climate but also to prevent habitat loss,” Brando concluded.

(Un)protected areas
The new research also highlights the crucial role of the currently existing network of protected areas and indigenous lands in the Amazon. Those areas can help buffer the loss of species diversity against future impacts.
“Even though [trees inside protected areas] are not totally immune to climate change, our models show that forests outside protected areas can lose up to a third more species,” said Gomes. “That’s why the preservation [of conserved areas] and the creation of corridors between them are so necessary, allowing biological dispersion and the migration of animals. Otherwise there will be only forest patches left in the Amazon.”
Vieira, of the Museu Paraense (linked with the Brazilian Ministry of Science, Technology, Innovation and Communications), said that it’s not difficult to imagine the most pessimistic study scenario becoming reality, given the trouble the world’s nations are having in achieving their carbon reduction goals as expressed in the 2015 Paris Agreement, as well as the difficulty Brazil has had in controlling deforestation, first under the Temer administration and now under the Bolsonaro administration .
June 2019 deforestation in the Brazilian Amazon increased by 88 percent as compared to the same month in 2018, and while this result is preliminary many analysts fear that 2019 could show a significant annual rise in deforestation.
However, the nation isn’t responding by curbing deforestation, rather its policies appear to be encouraging the opposite. In May, Brazilian Environment Minister Ricardo Salles proposed a reevaluation of all 334 federal conservation units , with an eye on reducing the size of some, and abolishing others. More recently he declared without offering any scientific evidence that Brazil has already reached “zero relative deforestation,” that is, deforestation in the Amazon represents, according to him, “0.002 percent of the biome.” Later he said the correct number is 0.16 percent.
Vieira responded: “There is a political scenario unfavorable to the environment in the country [of Brazil], with budget cuts in the environmental and scientific areas, reduction of operations in environmental control and inspection, setbacks in environmental legislation, freezing of indigenous lands demarcations and the threat of opening [indigenous reserves] to mining activities.
“To save the Amazon from destruction, initiatives should consider an integrated vision of the region, long-term public policies, respect for environmental legislation, as well as for indigenous territories and peoples, and there should be strong economic pressure to restrict the sale of products from deforested areas and protected areas of the Amazon,” Vieira concluded.
Gomes, V. H. F., Vieira, I. C. G., Salomão, R.P., & ter Steege, H. (2019). Amazonian tree species threatened by deforestation and climate change . Nature Climate Change , 9(7), 547-553.
FEEDBACK: Use this form to send a message to the author of this post. If you want to post a public comment, you can do that at the bottom of the page.

Special series
Forest trackers.
- Narco activity takes heavy toll on Colombia’s protected forests, satellite data show
- Photos confirm narcotraffickers operating in Peru’s Kakataibo Indigenous Reserve
- Bolivia’s El Curichi Las Garzas protected area taken over by land-grabbers
- Authorities struggle to protect Bolivian national park from drug-fueled deforestation
- Report: Illegal fishing and labor abuse rampant in China’s Indian Ocean fleet
- Restoring Indigenous aquaculture heals both ecosystems and communities in Hawai‘i
- Peru approves the creation of long-awaited marine protected area
- Thai plan to relax fishing law stokes fear of return to illegal catches, worker abuse

Amazon Conservation
- New bill to expand farmlands in the Amazon may derail Brazil’s green efforts
- Bird populations are mysteriously declining at an Amazon park in Ecuador & beyond
- New ban threatens traditional fishers in Brazil’s Mato Grosso state
- Deforestation haunts top Peruvian reserve and its Indigenous communities

Land rights and extractives
- Critics see payback in Indonesia’s plan to grant mining permits to religious groups
- Impunity and pollution abound in DRC mining along the road to the energy transition
- Women weave a culture of resistance and agroecology in Ecuador’s Intag Valley
- Hyundai ends aluminum deal with Adaro Minerals following K-pop protest

Endangered Environmentalists
- Mongabay video screening at Chile’s Supreme Court expected to help landmark verdict in Brazil
- Indonesian activist freed in hate speech case after flagging illegal shrimp farms
- Final cheetah conservationists freed in Iran, but the big cat’s outlook remains grim
- Indonesian activists face jail over FB posts flagging damage to marine park

Indonesia's Forest Guardians
- On a Borneo mountainside, Indigenous Dayak women hold fire and defend forest
- Fenced in by Sulawesi national park, Indigenous women make forestry breakout
- In Borneo, the ‘Power of Mama’ fight Indonesia’s wildfires with all-woman crew
- Pioneer agroforester Ermi, 73, rolls back the years in Indonesia’s Gorontalo

Conservation Effectiveness
- The conservation sector must communicate better (commentary)
- Thailand tries nature-based water management to adapt to climate change
- Forest restoration to boost biomass doesn’t have to sacrifice tree diversity
- How scientists and a community are bringing a Bornean river corridor back to life

Southeast Asian infrastructure
- Study: Indonesia’s new capital city threatens stable proboscis monkey population
- Indonesia’s new capital ‘won’t sacrifice the environment’: Q&A with Nusantara’s Myrna Asnawati Safitri
- Small farmers in limbo as Cambodia wavers on Tonle Sap conservation rules
- To build its ‘green’ capital city, Indonesia runs a road through a biodiverse forest

you're currently offline
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- My Bibliography
- Collections
- Citation manager
Save citation to file
Email citation, add to collections.
- Create a new collection
- Add to an existing collection
Add to My Bibliography
Your saved search, create a file for external citation management software, your rss feed.
- Search in PubMed
- Search in NLM Catalog
- Add to Search
Avian ecological succession in the Amazon: A long-term case study following experimental deforestation
Affiliations.
- 1 Biological Dynamics of Forest Fragments Project Instituto Nacional de Pesquisas da Amazônia (INPA) Manaus Brazil.
- 2 School of Renewable Natural Resources Louisiana State University and Louisiana State University AgCenter Baton Rouge LA USA.
- 3 Coleções Zoológicas - INPA Manaus Brazil.
- 4 Centre for Tropical Environmental and Sustainability Science College of Science and Engineering James Cook University Cairns Qld Australia.
- PMID: 31938486
- PMCID: PMC6953691
- DOI: 10.1002/ece3.5822
Approximately 20% of the Brazilian Amazon has now been deforested, and the Amazon is currently experiencing the highest rates of deforestation in a decade, leading to large-scale land-use changes. Roads have consistently been implicated as drivers of ongoing Amazon deforestation and may act as corridors to facilitate species invasions. Long-term data, however, are necessary to determine how ecological succession alters avian communities following deforestation and whether established roads lead to a constant influx of new species.We used data across nearly 40 years from a large-scale deforestation experiment in the central Amazon to examine the avian colonization process in a spatial and temporal framework, considering the role that roads may play in facilitating colonization.Since 1979, 139 species that are not part of the original forest avifauna have been recorded, including more secondary forest species than expected based on the regional species pool. Among the 35 species considered to have colonized and become established, a disproportionate number were secondary forest birds (63%), almost all of which first appeared during the 1980s. These new residents comprise about 13% of the current community of permanent residents.Widespread generalists associated with secondary forest colonized quickly following deforestation, with few new species added after the first decade, despite a stable road connection. Few species associated with riverine forest or specialized habitats colonized, despite road connection to their preferred source habitat. Colonizing species remained restricted to anthropogenic habitats and did not infiltrate old-growth forests nor displace forest birds.Deforestation and expansion of road networks into terra firme rainforest will continue to create degraded anthropogenic habitat. Even so, the initial pulse of colonization by nonprimary forest bird species was not the beginning of a protracted series of invasions in this study, and the process appears to be reversible by forest succession.
Keywords: Amazonia; Neotropics; colonization; deforestation; ecological species invasions; land‐use change; rain forest.
© 2019 The Authors. Ecology and Evolution published by John Wiley & Sons Ltd.
PubMed Disclaimer
Conflict of interest statement
None declared.
Study area, showing the Biological…
Study area, showing the Biological Dynamics of Forest Fragments Project (BDFFP; represented by…
The number of observed (gray…
The number of observed (gray bars) and expected (empty bars) bird species per…
Results of land cover classification…
Results of land cover classification as closed‐canopy forest (primary forest or mature regrowth;…
Similar articles
- Forest fires and deforestation in the central Amazon: Effects of landscape and climate on spatial and temporal dynamics. Dos Reis M, Graça PMLA, Yanai AM, Ramos CJP, Fearnside PM. Dos Reis M, et al. J Environ Manage. 2021 Jun 15;288:112310. doi: 10.1016/j.jenvman.2021.112310. Epub 2021 Mar 21. J Environ Manage. 2021. PMID: 33761331
- Land use change emission scenarios: anticipating a forest transition process in the Brazilian Amazon. Aguiar AP, Vieira IC, Assis TO, Dalla-Nora EL, Toledo PM, Santos-Junior RA, Batistella M, Coelho AS, Savaget EK, Aragão LE, Nobre CA, Ometto JP. Aguiar AP, et al. Glob Chang Biol. 2016 May;22(5):1821-40. doi: 10.1111/gcb.13134. Epub 2016 Feb 9. Glob Chang Biol. 2016. PMID: 26511401
- Amazon deforestation: A dangerous future indicated by patterns and trajectories in a hotspot of forest destruction in Brazil. Cabral BF, Yanai AM, Graça PMLA, Escada MIS, de Almeida CM, Fearnside PM. Cabral BF, et al. J Environ Manage. 2024 Mar;354:120354. doi: 10.1016/j.jenvman.2024.120354. Epub 2024 Feb 22. J Environ Manage. 2024. PMID: 38394876
- Climate change, allergy and asthma, and the role of tropical forests. D'Amato G, Vitale C, Rosario N, Neto HJC, Chong-Silva DC, Mendonça F, Perini J, Landgraf L, Solé D, Sánchez-Borges M, Ansotegui I, D'Amato M. D'Amato G, et al. World Allergy Organ J. 2017 Mar 7;10(1):11. doi: 10.1186/s40413-017-0142-7. eCollection 2017. World Allergy Organ J. 2017. PMID: 28286602 Free PMC article. Review.
- Beyond diversity loss and climate change: Impacts of Amazon deforestation on infectious diseases and public health. Ellwanger JH, Kulmann-Leal B, Kaminski VL, Valverde-Villegas JM, Veiga ABGD, Spilki FR, Fearnside PM, Caesar L, Giatti LL, Wallau GL, Almeida SEM, Borba MR, Hora VPD, Chies JAB. Ellwanger JH, et al. An Acad Bras Cienc. 2020 Apr 17;92(1):e20191375. doi: 10.1590/0001-3765202020191375. eCollection 2020. An Acad Bras Cienc. 2020. PMID: 32321030 Review.
- Species-specific traits affect bird species' susceptibility to global change. Rivas-Salvador J, Reif J. Rivas-Salvador J, et al. Naturwissenschaften. 2023 Nov 14;110(6):54. doi: 10.1007/s00114-023-01883-4. Naturwissenschaften. 2023. PMID: 37957333
- Climate-driven variation in dispersal ability predicts responses to forest fragmentation in birds. Weeks TL, Betts MG, Pfeifer M, Wolf C, Banks-Leite C, Barbaro L, Barlow J, Cerezo A, Kennedy CM, Kormann UG, Marsh CJ, Olivier PI, Phalan BT, Possingham HP, Wood EM, Tobias JA. Weeks TL, et al. Nat Ecol Evol. 2023 Jul;7(7):1079-1091. doi: 10.1038/s41559-023-02077-x. Epub 2023 May 29. Nat Ecol Evol. 2023. PMID: 37248334
- Range-wide sources of variation in reproductive rates of northern spotted owls. Rockweit JT, Jenkins JM, Hines JE, Nichols JD, Dugger KM, Franklin AB, Carlson PC, Kendall WL, Lesmeister DB, McCafferty C, Ackers SH, Andrews LS, Bailey LL, Burgher J, Burnham KP, Chestnut T, Conner MM, Davis RJ, Dilione KE, Forsman ED, Glenn EM, Gremel SA, Hamm KA, Herter DR, Higley JM, Horn RB, Lamphear DW, McDonald TL, Reid JA, Schwarz CJ, Simon DC, Sovern SG, Swingle JK, Wiens JD, Wise H, Yackulic CB. Rockweit JT, et al. Ecol Appl. 2023 Jan;33(1):e2726. doi: 10.1002/eap.2726. Epub 2022 Nov 30. Ecol Appl. 2023. PMID: 36053865 Free PMC article.
- Long-term changes in avian biomass and functional diversity within disturbed and undisturbed Amazonian rainforest. Luther DA, Cooper WJ, Jirinec V, Wolfe JD, Rutt CL, Bierregaard RO Jr, Lovejoy TE, Stouffer PC. Luther DA, et al. Proc Biol Sci. 2022 Aug 31;289(1981):20221123. doi: 10.1098/rspb.2022.1123. Epub 2022 Aug 17. Proc Biol Sci. 2022. PMID: 35975441 Free PMC article.
- Species richness response to human pressure hides important assemblage transformations. Cazalis V. Cazalis V. Proc Natl Acad Sci U S A. 2022 May 10;119(19):e2107361119. doi: 10.1073/pnas.2107361119. Epub 2022 May 2. Proc Natl Acad Sci U S A. 2022. PMID: 35500119 Free PMC article.
- Ahmed, S. E. , Ewers, R. M. , & Smith, M. J. (2013). Large scale spatio‐temporal patterns of road development in the Amazon rainforest. Environmental Conservation, 41(3), 253–264. 10.1017/s0376892913000520 - DOI
- Ahmed, S. E. , Lees, A. C. , Moura, N. G. , Gardner, T. A. , Barlow, J. , Ferreira, J. , & Ewers, R. M. (2014). Road networks predict human influence on Amazonian bird communities. Proceedings of the Royal Society B‐Biological Sciences, 281(1795), 20141742 10.1098/rspb.2014.1742 - DOI - PMC - PubMed
- Ahmed, S. E. , Souza, C. M. , Riberio, J. , & Ewers, R. M. (2013). Temporal patterns of road network development in the Brazilian Amazon. Regional Environmental Change, 13(5), 927–937. 10.1007/s10113-012-0397-z - DOI
- Altizer, S. , Bartel, R. , & Han, B. A. (2011). Animal migration and infectious disease risk. Science, 331(6015), 296–302. - PubMed
- Artaxo, P. (2019). Working together for Amazonia. Science, 363(6425), 323 10.1126/science.aaw6986 - DOI - PubMed
Associated data
- Dryad/10.5061/dryad.xpnvx0kb2
Related information
Linkout - more resources, full text sources.
- Europe PubMed Central
- PubMed Central

- Citation Manager
NCBI Literature Resources
MeSH PMC Bookshelf Disclaimer
The PubMed wordmark and PubMed logo are registered trademarks of the U.S. Department of Health and Human Services (HHS). Unauthorized use of these marks is strictly prohibited.
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Published: 01 September 2021
How deregulation, drought and increasing fire impact Amazonian biodiversity
- Xiao Feng ORCID: orcid.org/0000-0003-4638-3927 1 na1 ,
- Cory Merow 2 na1 ,
- Zhihua Liu ORCID: orcid.org/0000-0002-0086-5659 3 na1 ,
- Daniel S. Park ORCID: orcid.org/0000-0003-2783-530X 4 , 5 na1 ,
- Patrick R. Roehrdanz ORCID: orcid.org/0000-0003-4047-5011 6 na1 ,
- Brian Maitner ORCID: orcid.org/0000-0002-2118-9880 2 na1 ,
- Erica A. Newman 7 , 8 na1 ,
- Brad L. Boyle 7 , 9 ,
- Aaron Lien 8 , 10 ,
- Joseph R. Burger 7 , 8 , 11 ,
- Mathias M. Pires 12 ,
- Paulo M. Brando ORCID: orcid.org/0000-0001-8952-7025 13 , 14 , 15 ,
- Mark B. Bush ORCID: orcid.org/0000-0001-6894-8613 16 ,
- Crystal N. H. McMichael ORCID: orcid.org/0000-0002-1064-1499 17 ,
- Danilo M. Neves 18 ,
- Efthymios I. Nikolopoulos 19 ,
- Scott R. Saleska 7 ,
- Lee Hannah 6 ,
- David D. Breshears ORCID: orcid.org/0000-0001-6601-0058 10 ,
- Tom P. Evans ORCID: orcid.org/0000-0003-4591-1011 20 ,
- José R. Soto 10 ,
- Kacey C. Ernst 21 &
- Brian J. Enquist 7 , 22 na1
Nature volume 597 , pages 516–521 ( 2021 ) Cite this article
17k Accesses
62 Citations
662 Altmetric
Metrics details
- Biodiversity
- Biogeography
Biodiversity contributes to the ecological and climatic stability of the Amazon Basin 1 , 2 , but is increasingly threatened by deforestation and fire 3 , 4 . Here we quantify these impacts over the past two decades using remote-sensing estimates of fire and deforestation and comprehensive range estimates of 11,514 plant species and 3,079 vertebrate species in the Amazon. Deforestation has led to large amounts of habitat loss, and fires further exacerbate this already substantial impact on Amazonian biodiversity. Since 2001, 103,079–189,755 km 2 of Amazon rainforest has been impacted by fires, potentially impacting the ranges of 77.3–85.2% of species that are listed as threatened in this region 5 . The impacts of fire on the ranges of species in Amazonia could be as high as 64%, and greater impacts are typically associated with species that have restricted ranges. We find close associations between forest policy, fire-impacted forest area and their potential impacts on biodiversity. In Brazil, forest policies that were initiated in the mid-2000s corresponded to reduced rates of burning. However, relaxed enforcement of these policies in 2019 has seemingly begun to reverse this trend: approximately 4,253–10,343 km 2 of forest has been impacted by fire, leading to some of the most severe potential impacts on biodiversity since 2009. These results highlight the critical role of policy enforcement in the preservation of biodiversity in the Amazon.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
24,99 € / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
185,98 € per year
only 3,65 € per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others

A unifying modelling of multiple land degradation pathways in Europe
Climate change may reveal currently unavailable parts of species’ ecological niches

Curbing the major and growing threats from invasive alien species is urgent and achievable
Data availability.
The plant occurrences from the BIEN database are accessible using the RBIEN package ( https://github.com/bmaitner/RBIEN ). The climatic data are accessible from http://worldclim.org and the soil data are available from http://soilgrids.org . MODIS active fire and burned area products are available at http://modis-fire.umd.edu . The MODIS Vegetation Continuous Fields data are publicly available from https://lpdaac.usgs.gov/products/mod44bv006/ . The annual forest loss layers are available from http://earthenginepartners.appspot.com/science-2013-global-forest . The plant range maps are accessible at https://github.com/shandongfx/paper_Amazon_biodiversity_2021 . The vertebrate range maps are available from https://www.iucnredlist.org/resources/spatial-data-download . The SPEI data are available from SPEI Global Drought Monitor ( https://spei.csic.es/map ).
Code availability
The code to process the remote-sensing data is available at https://github.com/shandongfx/paper_Amazon_biodiversity_2021 .
Yachi, S. & Loreau, M. Biodiversity and ecosystem productivity in a fluctuating environment: the insurance hypothesis. Proc. Natl Acad. Sci. USA 96 , 1463–1468 (1999).
Article ADS CAS PubMed PubMed Central Google Scholar
Oliver, T. H. et al. Biodiversity and resilience of ecosystem functions. Trends Ecol. Evol. 30 , 673–684 (2015).
Article PubMed Google Scholar
Barlow, J., Berenguer, E., Carmenta, R. & França, F. Clarifying Amazonia’s burning crisis. Glob. Change Biol. 9 , 1 (2019).
Google Scholar
Brando, P. M. et al. The gathering firestorm in southern Amazonia. Sci. Adv. 6 , eaay1632 (2020).
IUCN. IUCN Red List of Threatened Species version 6.2 . https://www.iucnredlist.org/ (2019).
Flores, M. et al. WWF’s Living Amazon Initiative (Grambs Corporación Gráfica, 2010).
Hubbell, S. P. et al. How many tree species are there in the Amazon and how many of them will go extinct? Proc. Natl Acad. Sci. USA 105 Suppl. 1 , 11498–11504 (2008).
Nepstad, D. C., Stickler, C. M., Filho, B. S.- & Merry, F. Interactions among Amazon land use, forests and climate: prospects for a near-term forest tipping point. Philos. Trans. R. Soc. Lond. B 363 , 1737–1746 (2008).
Article Google Scholar
Rankin-de-Mérona, J. M. et al. Preliminary results of a large-scale tree inventory of upland rain forest in the Central Amazon. Acta Amazon. 22 , 493–534 (1992).
Sakschewski, B. et al. Resilience of Amazon forests emerges from plant trait diversity. Nat. Clim. Change 6 , 1032–1036 (2016).
Article ADS Google Scholar
Poorter, L. et al. Biomass resilience of Neotropical secondary forests. Nature 530 , 211–214 (2016).
Article ADS CAS PubMed Google Scholar
Beisner, B. E., Haydon, D. T. & Cuddington, K. Alternative stable states in ecology. Front. Ecol. Environ. 1 , 376–382 (2003).
Lovejoy, T. E. & Nobre, C. Amazon tipping point. Sci. Adv. 4 , eaat2340 (2018).
Article ADS PubMed PubMed Central Google Scholar
Veldman, J. W. Clarifying the confusion: old-growth savannahs and tropical ecosystem degradation. Philos. Trans. R. Soc. Lond. B 371 , (2016).
Arruda, D., Candido, H. G. & Fonseca, R. Amazon fires threaten Brazil’s agribusiness. Science 365 , 1387 (2019).
Article ADS PubMed CAS Google Scholar
Ter Steege, H. et al. Estimating the global conservation status of more than 15,000 Amazonian tree species. Sci. Adv. 1 , e1500936 (2015).
Gomes, V. H. F., Vieira, I. C. G., Salomão, R. P. & ter Steege, H. Amazonian tree species threatened by deforestation and climate change. Nat. Clim. Change 9 , 547–553 (2019).
Brando, P. et al. Amazon wildfires: scenes from a foreseeable disaster. Flora 268 , 151609 (2020).
Balch, J. K. et al. The susceptibility of southeastern Amazon forests to fire: insights from a large-scale burn experiment. Bioscience 65 , 893–905 (2015).
Barlow, J. et al. The critical importance of considering fire in REDD+ programs. Biol. Conserv. 154 , 1–8 (2012).
Cochrane, M. A. & Schulze, M. D. Fire as a recurrent event in tropical forests of the eastern Amazon: effects on forest structure, biomass, and species composition. Biotropica 31 , 2–16 (1999).
Brando, P. M. et al. Prolonged tropical forest degradation due to compounding disturbances: Implications for CO 2 and H 2 O fluxes. Glob. Change Biol. 25 , 2855–2868 (2019).
Barlow, J. & Peres, C. A. Fire-mediated dieback and compositional cascade in an Amazonian forest. Philos. Trans. R. Soc. Lond. B 363 , 1787–1794 (2008).
Cochrane, M. Tropical Fire Ecology: Climate Change, Land Use and Ecosystem Dynamics (Springer, 2010).
Uhl, C. & Kauffman, J. B. Deforestation, fire susceptibility, and potential tree responses to fire in the eastern Amazon. Ecology 71 , 437–449 (1990).
Cochrane, M. A. Fire science for rainforests. Nature 421 , 913–919 (2003).
Cochrane, M. A. & Laurance, W. F. Synergisms among fire, land use, and climate change in the Amazon. Ambio 37 , 522–527 (2008).
Nepstad, D. C. et al. Large-scale impoverishment of Amazonian forests by logging and fire. Nature 398 , 505–508 (1999).
Article ADS CAS Google Scholar
Aragão, L. E. O. C. et al. 21st Century drought-related fires counteract the decline of Amazon deforestation carbon emissions. Nat. Commun. 9 , 536 (2018).
Article ADS PubMed PubMed Central CAS Google Scholar
Nepstad, D. et al. Slowing Amazon deforestation through public policy and interventions in beef and soy supply chains. Science 344 , 1118–1123 (2014).
Hope, M. The Brazilian development agenda driving Amazon devastation. Lancet Planet. Health 3 , e409–e411 (2019).
Brown, J. H. On the relationship between abundance and distribution of species. Am. Nat. 124 , 255–279 (1984).
Barnagaud, J.-Y. et al. Ecological traits influence the phylogenetic structure of bird species co-occurrences worldwide. Ecol. Lett. 17 , 811–820 (2014).
Šímová, I. et al. Spatial patterns and climate relationships of major plant traits in the New World differ between woody and herbaceous species. J. Biogeogr. 45 , 895–916 (2018).
Enquist, B. J. et al. The commonness of rarity: Global and future distribution of rarity across land plants. Sci. Adv. 5 , eaaz0414 (2019).
May, P. H., Gebara, M. F., de Barcellos, L. M., Rizek, M. B. & Millikan, B. The Context of REDD+ in Brazil: Drivers, Agents, and Institutions, 3rd edition, https://doi.org/10.17528/cifor/006338 (Center for International Forestry Research, 2016).
Neves, D. M., Dexter, K. G., Baker, T. R., Coelho de Souza, F. & Oliveira-Filho, A. T. Evolutionary diversity in tropical tree communities peaks at intermediate precipitation. Sci. Rep. 10 , 1188 (2020).
Cadotte, M. W., Cardinale, B. J. & Oakley, T. H. Evolutionary history and the effect of biodiversity on plant productivity. Proc. Natl Acad. Sci. USA 105 , 17012–17017 (2008).
Hopkins, M. J. G. Modelling the known and unknown plant biodiversity of the Amazon Basin. J. Biogeogr. 34 , 1400–1411 (2007).
Wilson, E. O. in Biodiversity (eds Wilson E. O. & Peter F. M.) Ch. 1 (National Academies Press, 1988).
Brooks, T. M. et al. Habitat loss and extinction in the hotspots of biodiversity. Conserv. Biol. 16 , 909–923 (2002).
Gibbs, H. K. et al. Brazil’s soy moratorium. Science 347 , 377–378 (2015).
Alix-Garcia, J. & Gibbs, H. K. Forest conservation effects of Brazil’s zero deforestation cattle agreements undermined by leakage. Glob. Environ. Change 47 , 201–217 (2017).
Escobar, H. There’s no doubt that Brazil’s fires are linked to deforestation, scientists say. Science https://doi.org/10.1126/science.aaz2689 (2019).
Amazon fires: Brazil sends army to help tackle blazes. BBC News https://www.bbc.co.uk/news/world-latin-america-49452789 (24 August 2019).
Marengo, J. A., Tomasella, J., Soares, W. R., Alves, L. M. & Nobre, C. A. Extreme climatic events in the Amazon basin. Theor. Appl. Climatol. 107 , 73–85 (2012).
Malhi, Y. et al. Exploring the likelihood and mechanism of a climate-change-induced dieback of the Amazon rainforest. Proc. Natl Acad. Sci. USA 106 , 20610–20615 (2009).
Swann, A. L. S. et al. Continental-scale consequences of tree die-offs in North America: identifying where forest loss matters most. Environ. Res. Lett. 13 , 055014 (2018).
McCoy, T. Amazon fires dropped unexpectedly in September, after summer spike. Washington Post https://www.washingtonpost.com/world/the_americas/amazon-fires-dropped-unexpectedly-in-september-after-spiking-over-the-summer/2019/10/02/4ddc0026-e516-11e9-b403-f738899982d2_story.html (2 October 2019).
Moutinho, P., Guerra, R. & Azevedo-Ramos, C. Achieving zero deforestation in the Brazilian Amazon: what is missing? Elementa 4 , 000125 (2016).
Olson, D. M. et al. Terrestrial ecoregions of the world: a new map of life on Earth: A new global map of terrestrial ecoregions provides an innovative tool for conserving biodiversity. Bioscience 51 , 933–938 (2001).
Hansen, M. C. et al. High-resolution global maps of 21st-century forest cover change. Science 342 , 850–853 (2013).
Giglio, L., Schroeder, W. & Justice, C. O. The collection 6 MODIS active fire detection algorithm and fire products. Remote Sens. Environ. 178 , 31–41 (2016).
Giglio, L. MODIS Collection 6 Active Fire Product User’s Guide Revision A (Univ. Maryland, 2015).
Barlow, J., Lagan, B. O. & Peres, C. A. Morphological correlates of fire-induced tree mortality in a central Amazonian forest. J. Trop. Ecol. 19 , 291–299 (2003).
Brando, P. M. et al. Fire-induced tree mortality in a neotropical forest: the roles of bark traits, tree size, wood density and fire behavior. Glob. Change Biol. 18 , 630–641 (2012).
Gibbs, H. K. et al. Tropical forests were the primary sources of new agricultural land in the 1980s and 1990s. Proc. Natl Acad. Sci. USA 107 , 16732–16737 (2010).
Barlow, J. & Peres, C. in Emerging Threats to Tropical Forests (eds. Laurance, W. F. & Peres, C. A.) 225–240 (Univ. Chicago Press, 2006).
Barlow, J. et al. Wildfires in bamboo-dominated Amazonian forest: impacts on above-ground biomass and biodiversity. PLoS ONE 7 , e33373 (2012).
Gerwing, J. J. Degradation of forests through logging and fire in the eastern Brazilian Amazon. For. Ecol. Manage. 157 , 131–141 (2002).
Brando, P. M. et al. Abrupt increases in Amazonian tree mortality due to drought-fire interactions. Proc. Natl Acad. Sci. USA 111 , 6347–6352 (2014).
Barlow, J. & Peres, C. A. Avifaunal responses to single and recurrent wildfires in Amazonian forests. Ecol. Appl. 14 , 1358–1373 (2004).
Paolucci, L. N., Schoereder, J. H., Brando, P. M. & Andersen, A. N. Fire-induced forest transition to derived savannas: cascading effects on ant communities. Biol. Conserv. 214 , 295–302 (2017).
Roy, D. P. & Kumar, S. S. Multi-year MODIS active fire type classification over the Brazilian Tropical Moist Forest Biome. Int. J. Digital Earth 10 , 54–84 (2017).
Giglio, L., Schroeder, W., Hall, J. V. & Justice, C. O. MODIS Collection 6 Active Fire Product User’s Guide Revision B (Univ. Maryland, 2018).
Barriopedro, D., Fischer, E. M., Luterbacher, J., Trigo, R. M. & García-Herrera, R. The hot summer of 2010: redrawing the temperature record map of Europe. Science 332 , 220–224 (2011).
Chen, Y. et al. Forecasting fire season severity in South America using sea surface temperature anomalies. Science 334 , 787–791 (2011).
Giglio, L. et al. Assessing variability and long-term trends in burned area by merging multiple satellite fire products. Biogeosciences 7 , 1171–1186 (2010).
Justice, C. O. et al. The MODIS fire products. Remote Sens. Environ. 83 , 244–262 (2002).
Giglio, L., Boschetti, L., Roy, D. P., Humber, M. L. & Justice, C. O. The Collection 6 MODIS burned area mapping algorithm and product. Remote Sens. Environ. 217 , 72–85 (2018).
Nóbrega, C. C., Brando, P. M., Silvério, D. V., Maracahipes, L. & de Marco, P. Effects of experimental fires on the phylogenetic and functional diversity of woody species in a neotropical forest. For. Ecol. Manage. 450 , 117497 (2019).
Alencar, A., Nepstad, D. & Diaz, M. C. V. Forest understory fire in the Brazilian Amazon in ENSO and Non-ENSO years: area burned and committed carbon emissions. Earth Interact. 10 , 1–17 (2006).
Siegert, F., Ruecker, G., Hinrichs, A. & Hoffmann, A. A. Increased damage from fires in logged forests during droughts caused by El Niño. Nature 414 , 437–440 (2001).
Cochrane, M. A. & Laurance, W. F. Fire as a large-scale edge effect in Amazonian forests. J. Trop. Ecol. 18 , 311–325 (2002).
Ray, D., Nepstad, D. & Moutinho, P. Micrometeorological and canopy controls of fire susceptibility in a forested Amazon landscape. Ecol. Appl. 15 , 1664–1678 (2005).
Silvério, D. V. et al. Fire, fragmentation, and windstorms: a recipe for tropical forest degradation. J. Ecol. 107 , 656–667 (2019).
Guisan, A. & Zimmermann, N. E. Predictive habitat distribution models in ecology. Ecol. Modell. 135 , 147–186 (2000).
Fegraus, E. Tropical Ecology Assessment and Monitoring Network (TEAM Network). Biodivers. Ecol. 4 , 287–287 (2012).
Peet, R. K., Lee, M. T., Jennings, M. D. & Faber-Langendoen, D. VegBank: a permanent, open-access archive for vegetation plot data. Biodivers. Ecol. 4 , 233–241 (2012).
DeWalt, S. J., Bourdy, G., Chavez de Michel, L. R. & Quenevo, C. Ethnobotany of the Tacana: quantitative inventories of two permanent plots of Northwestern Bolivia. Econ. Bot. 53 , 237–260 (1999).
USDA Forest Service. Forest Inventory and Analysis National Program, http://www.fia.fs.fed.us/ (2013).
Wiser, S. K., Bellingham, P. J. & Burrows, L. E. Managing biodiversity information: development of New Zealand’s National Vegetation Survey databank. N. Z. J. Ecol. 25 , 1–17 (2001).
Anderson-Teixeira, K. J. et al. CTFS-ForestGEO: a worldwide network monitoring forests in an era of global change. Glob. Change Biol. 21 , 528–549 (2015).
Enquist, B. & Boyle, B. SALVIAS – the SALVIAS vegetation inventory database. Biodivers. Ecol. 4 , 288 (2012).
GBIF.org. GBIF Occurrence Download https://doi.org/10.15468/dl.yubndf (2018).
Dauby, G. et al. RAINBIO: a mega-database of tropical African vascular plants distributions. PhytoKeys 74 , 1–18 (2016).
Arellano, G. et al. A standard protocol for woody plant inventories and soil characterisation using temporary 0.1-ha plots in tropical forests. J. Trop. For. Sci. 28 , 508–516 (2016).
O’Connell, B. M. et al. The Forest Inventory and Analysis Database: Database Description and User Guide for Phase 2 (version 6.1), https://doi.org/10.2737/fs-fiadb-p2-6.1 (USDA Forest Service, 2016).
Oliveira-Filho, A. T. NeoTropTree, Flora arbórea da Região Neotropical: Um Banco de Dados Envolvendo Biogeografia, Diversidade e Conservação, http://www.neotroptree.info (Univ. Federal de Minas Gerais, 2017).
Peet, R. K., Lee, M. T., Jennings, M. D. & Faber-Langendoen, D. VegBank: The Vegetation Plot Archive of the Ecological Society of America , http://vegbank.org (accessed 2013).
Boyle, B. et al. The taxonomic name resolution service: an online tool for automated standardization of plant names. BMC Bioinf. 14 , 16 (2013).
Goldsmith, G. R. et al. Plant-O-Matic: a dynamic and mobile guide to all plants of the Americas. Methods Ecol. Evol. 7 , 960–965 (2016).
McFadden, I. R. et al. Temperature shapes opposing latitudinal gradients of plant taxonomic and phylogenetic β diversity. Ecol. Lett. 22 , 1126–1135 (2019).
Enquist, B. J., Condit, R., Peet, R. K., Schildhauer, M. & Thiers, B. M. Cyberinfrastructure for an integrated botanical information network to investigate the ecological impacts of global climate change on plant biodiversity. Preprint at https://doi.org/10.7287/peerj.preprints.2615v2 (2016).
Maitner, B. S. et al. The BIEN R package: A tool to access the Botanical Information and Ecology Network (BIEN) database. Methods Ecol. Evol. 9 , 373–379 (2017).
Phillips, S. J. & Dudik, M. Modeling of species distributions with Maxent: new extensions and a comprehensive evaluation. Ecography 31 , 161–175 (2008).
Merow, C. & Silander, J. A. A comparison of Maxlike and Maxent for modelling species distributions. Methods Ecol. Evol. 5 , 215–225 (2014).
Aiello-Lammens, M. E., Boria, R. A., Radosavljevic, A., Vilela, B. & Anderson, R. P. spThin: an R package for spatial thinning of species occurrence records for use in ecological niche models. Ecography 38 , 541–545 (2015).
Grubbs, F. E. Sample criteria for testing outlying observations. Ann. Math. Statist. 21 , 27–58 (1950).
Article MathSciNet MATH Google Scholar
Komsta, L. outliers: Tests for outliers. R package v.0.14 (2011).
Fick, S. E. & Hijmans, R. J. WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 37 , 4302–4315 (2017).
Hijmans, R. J., Cameron, S. E., Parra, J. L., Jones, P. G. & Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25 , 1965–1978 (2005).
Mueller-Dombois, D. & Ellenberg, H. Aims and Methods of Vegetation Ecology (Wiley, 1974).
Friedman, J., Hastie, T. & Tibshirani, R. glmnet: Lasso and elastic-net regularized generalized linear models. R package v.4.0-2 (2020).
Phillips, S. J., Anderson, R. P. & Schapire, R. E. Maximum entropy modeling of species geographic distributions. Ecol. Modell. 190 , 231–259 (2006).
Drake, J. M. Range bagging: a new method for ecological niche modelling from presence-only data. J. R. Soc. Interface 12 , 20150086 (2015).
Article PubMed PubMed Central Google Scholar
Cardoso, D. et al. Amazon plant diversity revealed by a taxonomically verified species list. Proc. Natl Acad. Sci. USA 114 , 10695–10700 (2017).
Article CAS PubMed PubMed Central Google Scholar
Warton, D. I. & Shepherd, L. C. Poisson point process models solve the “pseudo-absence problem” for presence-only data in ecology. Ann. Appl. Stat. 4 , 1383–1402 (2010).
MathSciNet MATH Google Scholar
Renner, I. W. et al. Point process models for presence-only analysis. Methods Ecol. Evol. 6 , 366–379 (2015).
Dinerstein, E. et al. An ecoregion-based approach to protecting half the terrestrial realm. Bioscience 67 , 534–545 (2017).
Roberts, D. R. et al. Cross-validation strategies for data with temporal, spatial, hierarchical, or phylogenetic structure. Ecography 40 , 913–929 (2016).
Phillips, S. J. Transferability, sample selection bias and background data in presence-only modelling: a response to Peterson et al . (2007). Ecography 31 , 272–278 (2008).
Merow, C., Smith, M. J. & Silander, J. A. Jr. A practical guide to MaxEnt for modeling species’ distributions: what it does, and why inputs and settings matter. Ecography 36 , 1058–1069 (2013).
Qiao, H. et al. An evaluation of transferability of ecological niche models. Ecography 42 , 521–534 (2019).
Peterson, A. T., Papeş, M. & Soberón, J. Rethinking receiver operating characteristic analysis applications in ecological niche modeling. Ecol. Modell. 213 , 63–72 (2008).
Allouche, O., Tsoar, A. & Kadmon, R. Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). J. Appl. Ecol. 43 , 1223–1232 (2006).
Jung, M. et al. Areas of global importance for terrestrial biodiversity, carbon, and water. Preprint at https://doi.org/10.1101/2020.04.16.021444 (2020).
Carlson, C. J. et al. Climate change will drive novel cross-species viral transmission. Preprint at https://doi.org/10.1101/2020.01.24.918755 (2020).
BirdLife International. IUCN Red List for Birds http://www.birdlife.org (2019).
Brooks, T. M. et al. Measuring terrestrial area of habitat (AOH) and its utility for the IUCN Red List. Trends Ecol. Evol. 34 , 977–986 (2019).
de Area Leão Pereira, E. J., de Santana Ribeiro, L. C., da Silva Freitas, L. F. & de Barros Pereira, H. B. Brazilian policy and agribusiness damage the Amazon rainforest. Land Use Policy 92 , 104491 (2020).
Garcia, R. T. After Brazil’s summer of fire, the militarization of the Amazon remains. Foreign Policy https://foreignpolicy.com/2019/11/19/militarization-amazon-legacy-brazil-forest-fire-bolsonaro/ (19 November 2019).
Vicente-Serrano, S. M., Beguería, S. & López-Moreno, J. I. A multiscalar drought index sensitive to global warming: The Standardized Precipitation Evapotranspiration Index. J. Clim. 23 , 1696–1718 (2010).
Feldpausch, T. R. et al. Amazon forest response to repeated droughts. Global Biogeochem. Cycles 30 , 964–982 (2016).
Marin, P.-G., Julio, C. J., Arturo, R.-T. D. & Jose, V.-N. D. Drought and spatiotemporal variability of forest fires across Mexico. Chin. Geogr. Sci. 28 , 25–37 (2018).
Adams, H. D. et al. Temperature sensitivity of drought-induced tree mortality portends increased regional die-off under global-change-type drought. Proc. Natl Acad. Sci. USA 106 , 7063–7066 (2009).
Download references
Acknowledgements
We acknowledge the herbaria that contributed data to this work: HA, FCO, MFU, UNEX, VDB, ASDM, BPI, BRI, CLF, L, LPB, AD, TAES, FEN, FHO, A, ANSM, BCMEX, RB, TRH, AAH, ACOR, AJOU, UI, AK, ALCB, AKPM, EA, AAU, ALU, AMES, AMNH, AMO, ANA, GH, ARAN, ARM, AS, CICY, ASU, BAI, AUT, B, BA, BAA, BAB, BACP, BAF, BAL, COCA, BARC, BBS, BC, BCN, BCRU, BEREA, BG, BH, BIO, BISH, SEV, BLA, BM, MJG, BOL, CVRD, BOLV, BONN, BOUM, BR, BREM, BRLU, BSB, BUT, C, CAMU, CAN, CANB, CAS, CAY, CBG, CBM, CEN, CEPEC, CESJ, CHR, ENCB, CHRB, CIIDIR, CIMI, CLEMS, COA, COAH, COFC, CP, COL, COLO, CONC, CORD, CPAP, CPUN, CR, CRAI, FURB, CU, CRP, CS, CSU, CTES, CTESN, CUZ, DAO, HB, DAV, DLF, DNA, DS, DUKE, DUSS, E, HUA, EAC, ECU, EIF, EIU, GI, GLM, GMNHJ, K, GOET, GUA, EKY, EMMA, HUAZ, ERA, ESA, F, FAA, FAU, UVIC, FI, GZU, H, FLAS, FLOR, HCIB, FR, FTG, FUEL, G, GB, GDA, HPL, GENT, GEO, HUAA, HUJ, CGE, HAL, HAM, IAC, HAMAB, HAS, HAST, IB, HASU, HBG, IBUG, HBR, IEB, HGI, HIP, IBGE, ICEL, ICN, ILL, SF, NWOSU, HO, HRCB, HRP, HSS, HU, HUAL, HUEFS, HUEM, HUSA, HUT, IAA, HYO, IAN, ILLS, IPRN, FCQ, ABH, BAFC, BBB, INPA, IPA, BO, NAS, INB, INEGI, INM, MW, EAN, IZTA, ISKW, ISC, GAT, IBSC, UCSB, ISU, IZAC, JBAG, JE, SD, JUA, JYV, KIEL, ECON, TOYA, MPN, USF, TALL, RELC, CATA, AQP, KMN, KMNH, KOR, KPM, KSTC, LAGU, UESC, GRA, IBK, KTU, KU, PSU, KYO, LA, LOMA, SUU, UNITEC, NAC, IEA, LAE, LAF, GMDRC, LCR, LD, LE, LEB, LI, LIL, LINN, AV, HUCP, MBML, FAUC, CNH, MACF, CATIE, LTB, LISI, LISU, MEXU, LL, LOJA, LP, LPAG, MGC, LPD, LPS, IRVC, MICH, JOTR, LSU, LBG, WOLL, LTR, MNHN, CDBI, LYJB, LISC, MOL, DBG, AWH, NH, HSC, LMS, MELU, NZFRI, M, MA, UU, UBT, CSUSB, MAF, MAK, MB, KUN, MARY, MASS, MBK, MBM, UCSC, UCS, JBGP, OBI, BESA, LSUM, FULD, MCNS, ICESI, MEL, MEN, TUB, MERL, CGMS, FSU, MG, HIB, TRT, BABY, ETH, YAMA, SCFS, SACT, ER, JCT, JROH, SBBG, SAV, PDD, MIN, SJSU, MISS, PAMP, MNHM, SDSU, BOTU, MPU, MSB, MSC, CANU, SFV, RSA, CNS, JEPS, BKF, MSUN, CIB, VIT, MU, MUB, MVFA, SLPM, MVFQ, PGM, MVJB, MVM, MY, PASA, N, HGM, TAM, BOON, MHA, MARS, COI, CMM, NA, NCSC, ND, NU, NE, NHM, NHMC, NHT, UFMA, NLH, UFRJ, UFRN, UFS, ULS, UNL, US, NMNL, USP, NMR, NMSU, XAL, NSW, ZMT, BRIT, MO, NCU, NY, TEX, U, UNCC, NUM, O, OCLA, CHSC, LINC, CHAS, ODU, OKL, OKLA, CDA, OS, OSA, OSC, OSH, OULU, OXF, P, PACA, PAR, UPS, PE, PEL, SGO, PEUFR, PH, PKDC, SI, PMA, POM, PORT, PR, PRC, TRA, PRE, PY, QMEX, QCA, TROM, QCNE, QRS, UH, R, REG, RFA, RIOC, RM, RNG, RYU, S, SALA, SANT, SAPS, SASK, SBT, SEL, SING, SIU, SJRP, SMDB, SNM, SOM, SP, SRFA, SPF, STL, STU, SUVA, SVG, SZU, TAI, TAIF, TAMU, TAN, TEF, TENN, TEPB, TI, TKPM, TNS, TO, TU, TULS, UADY, UAM, UAS, UB, UC, UCR, UEC, UFG, UFMT, UFP, UGDA, UJAT, ULM, UME, UMO, UNA, UNM, UNR, UNSL, UPCB, UPNA, USAS, USJ, USM, USNC, USZ, UT, UTC, UTEP, UV, VAL, VEN, VMSL, VT, W, WAG, WII, WELT, WIS, WMNH, WS, WTU, WU, Z, ZSS, ZT, CUVC, AAS, AFS, BHCB, CHAM, FM, PERTH and SAN. X.F., D.S.P., E.A.N., A.L. and J.R.B. were supported by the University of Arizona Bridging Biodiversity and Conservation Science program. Z.L. was supported by NSFC (41922006) and K. C. Wong Education Foundation. The BIEN working group was supported by the National Center for Ecological Analysis and Synthesis, a centre funded by NSF EF-0553768 at the University of California, Santa Barbara, and the State of California. Additional support for the BIEN working group was provided by iPlant/Cyverse via NSF DBI-0735191. B.J.E., B.M. and C.M. were supported by NSF ABI-1565118. B.J.E. and C.M. were supported by NSF ABI-1565118 and NSF HDR-1934790. B.J.E., L.H. and P.R.R. were supported by the Global Environment Facility SPARC project grant (GEF-5810). D.D.B. was supported in part by NSF DEB-1824796 and NSF DEB-1550686. S.R.S. was supported by NSF DEB-1754803. X.F. and A.L. were partly supported by NSF DEB-1824796. B.J.E. and D.M.N. were supported by NSF DEB-1556651. M.M.P. is supported by the São Paulo Research Foundation (FAPESP), grant 2019/25478-7. D.M.N. was supported by Instituto Serrapilheira/Brazil (Serra-1912-32082). E.I.N. was supported by NSF HDR-1934712. We thank L. López-Hoffman and L. Baldwin for constructive comments.
Author information
These authors contributed equally: Xiao Feng, Cory Merow, Zhihua Liu, Daniel S. Park, Patrick R. Roehrdanz, Brian Maitner, Erica A. Newman, Brian J. Enquist
Authors and Affiliations
Department of Geography, Florida State University, Tallahassee, FL, USA
Eversource Energy Center and Department of Ecology and Evolutionary Biology, University of Connecticut, Storrs, CT, USA
Cory Merow & Brian Maitner
CAS Key Laboratory of Forest Ecology and Management, Institute of Applied Ecology, Chinese Academy of Sciences, Shenyang, China
Department of Biological Sciences, Purdue University, West Lafayette, IN, USA
Daniel S. Park
Purdue Center for Plant Biology, Purdue University, West Lafayette, IN, USA
The Moore Center for Science, Conservation International, Arlington, VA, USA
Patrick R. Roehrdanz & Lee Hannah
Department of Ecology and Evolutionary Biology, University of Arizona, Tucson, AZ, USA
Erica A. Newman, Brad L. Boyle, Joseph R. Burger, Scott R. Saleska & Brian J. Enquist
Arizona Institutes for Resilience, University of Arizona, Tucson, AZ, USA
Erica A. Newman, Aaron Lien & Joseph R. Burger
Hardner & Gullison Associates, Amherst, NH, USA
Brad L. Boyle
School of Natural Resources and the Environment, University of Arizona, Tucson, AZ, USA
Aaron Lien, David D. Breshears & José R. Soto
Department of Biology, University of Kentucky, Lexington, KY, USA
Joseph R. Burger
Departamento de Biologia Animal, Universidade Estadual de Campinas, Campinas, Brazil
Mathias M. Pires
Department of Earth System Science, University of California, Irvine, Irvine, CA, USA
Paulo M. Brando
Woodwell Climate Research Center, Falmouth, MA, USA
Instituto de Pesquisa Ambiental da Amazônia (IPAM), Brasilia, Brazil
Insitute for Global Ecology, Florida Institute of Technology, Melbourne, FL, USA
Mark B. Bush
Department of Ecosystem and Landscape Dynamics, Institute for Biodiversity and Ecosystem Dynamics, University of Amsterdam, Amsterdam, The Netherlands
Crystal N. H. McMichael
Institute of Biological Sciences, Federal University of Minas Gerais, Belo Horizonte, Brazil
Danilo M. Neves
Department of Mechanical and Civil Engineering, Florida Institute of Technology, Melbourne, FL, USA
Efthymios I. Nikolopoulos
School of Geography, Development and Environment, University of Arizona, Tucson, AZ, USA
Tom P. Evans
Department of Epidemiology and Biostatistics, College of Public Health, University of Arizona, Tucson, AZ, USA
Kacey C. Ernst
The Santa Fe Institute, Santa Fe, NM, USA
Brian J. Enquist
You can also search for this author in PubMed Google Scholar
Contributions
X.F. conceived the idea, which was refined by discussion with D.S.P., C.M., B.M., P.R.R., E.A.N., B.L.B., A.L., J.R.B., D.D.B., J.R.S., K.C.E. and B.J.E.; X.F. and Z.L. processed the remote-sensing data; C.M., X.F., B.M., B.L.B., D.S.P. and B.J.E. conducted the analyses of plant data; P.R.R., C.M., B.M., X.F. and D.S.P. conducted the analyses of vertebrate data; X.F., C.M., S.R.S. and E.A.N. processed the drought data; D.S.P., X.F., C.M., P.R.R. and B.M. designed the illustrations with help from B.J.E., D.D.B., K.C.E. and E.A.N.; E.A.N., X.F., and D.S.P. conducted the statistical analyses with help from B.J.E.; X.F., B.J.E., B.M., A.L., J.R.B., D.S.P., C.M., E.A.N., Z.L. and P.R.R. wrote the original draft; all authors contributed to interpreting the results and the editing of manuscript drafts. B.J.E., C.M., K.C.E. and D.D.B. led to the acquisition of the financial support for the project. X.F., C.M., B.M., D.S.P., P.R.R., Z.L., E.A.N. and B.J.E. contributed equally to data, analyses and writing.
Corresponding author
Correspondence to Xiao Feng .
Ethics declarations
Competing interests.
The authors declare no competing interests.
Additional information
Peer review information Nature thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended data fig. 1 fire-impacted forest and forest loss in the amazon basin..
a – h , Visualization of fire-impacted forest ( a , b ), forest loss without fire ( c , d ), fire-impacted forest with forest loss ( e , f ), and fire-impacted forest without forest loss ( g , h ) in the Amazon Basin based on MODIS burned area (left panels) and active fire (right panels). Data in a – d are resampled from the 500m (MODIS burned area) or 1 km (MODIS active fire) to 10 km resolution using mean function and thresholded at 0.01 to illustrate the temporal dynamics. Black represents non-forested areas masked out from this study. The cumulative fire-impacted forest is classified into two categories: fire-impacted forest with forest loss ( e , f ) and fire-impacted forest without forest loss ( g , h ). Data in e – h are resampled to 10 km using mean function to illustrate the cumulative percentages of impacts.
Extended Data Fig. 2 Scatter plot of species’ range impacted by fire.
Scatter plot of species’ range size in Amazon forest (x-axis) and percentage of total range impacted by fire (red) and forest loss without fire (black) up to 2019 for plants (left panel) and vertebrates (right panel).
Extended Data Fig. 3 Density plot of species’ cumulative range impacted by fire.
Density plot of species’ cumulative range impacted by fire. The different colours represent years 2001-2019. The x-axis is log10 transformed.
Extended Data Fig. 4 Summary of forest impacts in the Amazon Basin.
Areas of forest impact in the Amazon Basin estimated from MODIS burned area (top) and MODIS active fire (bottom).
Extended Data Fig. 5 Cumulative impacts on biodiversity in the Amazon Basin.
Cumulative effects of forest loss without fire on biodiversity in the Amazon rainforest. In the left panels, the black and grey shading represent the cumulative forest loss without fire based on MODIS burned area and MODIS active fire, respectively. Coloured areas represent the lower and upper bounds of cumulative numbers of a , plant and c , vertebrate species’ ranges impacted. Right panels depict the relationships between the cumulative forest loss without fire (based on MODIS burned area) and cumulative number of b , plant and d , vertebrate species. Coloured lines represent predicted values of an ordinary least squares linear regression and grey bands define the two-sided 95% confidence interval (two-sided, p values = 0.00). The silhouette of the tree is from http://phylopic.org/ ; silhouette of the monkey is courtesy of Mathias M. Pires.
Extended Data Fig. 6 Fire-impacted forest in Brazil.
Newly fire-impacted forest in Brazil (based on MODIS active fire). a shows the area of fire-impacted forest not explained by drought conditions. Different colours represent years from different policy regimes: pre-regulations in light red (mean value in dark red), regulation in grey (mean value in black dashed line), and 2019 in blue. The y-axis represents the difference between actual area and area predicted by drought conditions calibrated by data from regulation years ( Methods ). A positive value on the y-axis represents more area than expected, using the regulation years as a baseline. b shows a scatter plot of newly fire-impacted forest in Brazil and drought conditions (SPEI); The lines represent the ordinary least squares linear regression between fire-impacted forest and drought conditions for pre-regulation (red) and regulation (black) respectively.
Extended Data Fig. 7 Fire-impacted forest in different countries.
The contribution (0–1) of different countries to the newly fire-impacted forest each year based on MODIS active fire (top) and MODIS burned area (bottom).
Extended Data Figure 8 Impacts of fire on forest and biodiversity in Brazil.
a , Newly fire-impacted forest, b , new range impact on plants and c , new range impacts on vertebrate species in Brazil each year (based on MODIS active fire) that are not predicted by drought conditions. The colours represent three policy regimes: pre-regulation in red, regulation in grey and 2019 in blue. The y-axis represents the difference between actual value (area or range impacted by fire) and the values predicted by drought conditions calibrated by data from regulation years ( Methods ). A positive value on the y-axis represents more area or range impacted by fire than the expectation using the regulation years as a baseline. The dotted lines represent a smooth curve fitted to the values based on the loess method.
Supplementary information
‘supplementary discussion’ and ‘this file contains the supplementary discussion’, reporting summary, rights and permissions.
Reprints and permissions
About this article
Cite this article.
Feng, X., Merow, C., Liu, Z. et al. How deregulation, drought and increasing fire impact Amazonian biodiversity. Nature 597 , 516–521 (2021). https://doi.org/10.1038/s41586-021-03876-7
Download citation
Received : 22 November 2019
Accepted : 04 August 2021
Published : 01 September 2021
Issue Date : 23 September 2021
DOI : https://doi.org/10.1038/s41586-021-03876-7
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
This article is cited by
Variation of floristic diversity, community composition, endemism, and conservation status of tree species in tropical rainforests of sri lanka across a wide altitudinal gradient.
- Nimalka Sanjeewani
- Dilum Samarasinghe
- Janendra De Costa
Scientific Reports (2024)
Comparative analysis of seed biomass from Amazonian fruits for activated carbon production
- Edina Ruth Mendes Leal Mafra
- Thiago de Paula Protásio
- Lina Bufalino
Biomass Conversion and Biorefinery (2024)
Assessing changes in global fire regimes
- Sayedeh Sara Sayedi
- Benjamin W. Abbott
- Anne-Laure Daniau
Fire Ecology (2024)
The role of high-biodiversity regions in preserving Nature’s Contributions to People
- Marta Cimatti
- Rebecca Chaplin-Kramer
- Moreno Di Marco
Nature Sustainability (2023)
Forest conservation in Indigenous territories and protected areas in the Brazilian Amazon
- Yuanwei Qin
- Xiangming Xiao
- Philip Martin Fearnside
By submitting a comment you agree to abide by our Terms and Community Guidelines . If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.
An official website of the United States government
Here's how you know
Official websites use .gov A .gov website belongs to an official government organization in the United States.
Secure .gov websites use HTTPS. A lock ( Lock Locked padlock ) or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.

- Search Awards
- Recent Awards
- Presidential and Honorary Awards
- About Awards
- How to Manage Your Award
- Grant General Conditions
- Cooperative Agreement Conditions
- Special Conditions
- Federal Demonstration Partnership
- Policy Office Website
| | |
| April 24, 2015 | |
| April 24, 2015 | |
| 1541489 | |
| Standard Grant | |
| Thomas Baerwald BCS �Division Of Behavioral and Cognitive Sci SBE �Direct For Social, Behav & Economic Scie | |
| September 1, 2014 | |
| December 31, 2016�(Estimated) | |
| $3,559.00 | |
| $3,559.00 | |
| Simmons | |
| 1523 UNION RD RM 207 GAINESVILLE FL �US �32611-1941 (352)392-3516 | |
| FL �US �32611-2002 | |
| Geography and Spatial Sciences | |
| 4900 | |
| 4900 | |
| 47.075 |
Note: When clicking on a Digital Object Identifier (DOI) number, you will be taken to an external site maintained by the publisher. Some full text articles may not yet be available without a charge during the embargo (administrative interval). Some links on this page may take you to non-federal websites. Their policies may differ from this site.
This Project Outcomes Report for the General Public is displayed verbatim as submitted by the Principal Investigator (PI) for this award. Any opinions, findings, and conclusions or recommendations expressed in this Report are those of the PI and do not necessarily reflect the views of the National Science Foundation; NSF has not approved or endorsed its content.
Motivation. Scientists have identified a number of factors to explain Amazonian deforestation. This project examines another driver that has not been adequately studied, namely that of contentious social interactions and conflict over access to land resources on forest cover, referred to here as Contentious Land Change (CLC). Land conflict, globally associated with agrarian reform, has waxed and waned in the Brazilian Amazon over the past three decades, a period that witnessed high rates of deforestation and hundreds of land conflict related fatalities.
The nexus between land conflict and natural resource impact is a widespread phenomenon. Transnational social movements like the Movement of the Landless Rural Workers (MST) and La Via Campesina have recently stimulated the rural poor to question access to land, often limited for institutional and historical reasons at odds with social equity. This has motivated a rural constituency that aggressively seeks more dignified, farm-based livelihoods. As a consequence, contention over land has seized headlines throughout the Global South, with notable movements in South American, Southeast Asia and Africa.
Our examination of CLC focuses on the Brazil-Nut Polygon, or BNP of southeastern Amazonia (Figure 1). We employ a spatial analytic model using panel data at the property level generated from unique sources, including (1) a multi-decadal satellite image series covering 27 years; (2) cadastral maps for 180 large holdings (4,886 ha average) and agrarian reform settlements,; and (3) a newspaper archive detailing land conflicts unfolding at property scale for the same period as the satellite imagery (~8,000 pages). This information enables the systematic evaluation of deforestation associated with the social inequalities driving land conflict in the Amazon Basin.
Conceptual Framework . This study dispenses with the LCS presumption that landholdings are managed by unitary decision-makers acting in the interest of market production. Often, deforestation results from contentious interactions between powerful elites and the rural poor, groups with differing motivations and agricultural practices. An important component of land change in forest frontiers results as land managers deforest in order to engage in agricultural activities, a process we refer to as agronomic land change, or ALC. Nevertheless, the presence of land conflict alters this dynamic by introducing new behaviors and social processes that do not conform to a static, rational actor model. Changes in land cover in contentious settings stem from a variety of actions taken by competing claimants. These include preemptive deforestation in anticipation of seizure by alternative claimants, excessive land clearance meant to stake a claim, the use of fire as a conflict weapon, and resource mining (valuable hardwoods, soils) in advance of expected dispossession .
For Amazonia, we hypothesize that deforestation reflects both ALC and CLC, and that CLC adds to the total amount of deforestation than would otherwise occur under ALC. This is depicted in Figure 2 for two hypothetical holdings of 4,000 ha. One holding experiences CLC and the other does not. Both follow a linear land clearance pattern at first, a property “life cycle”. Divergence sets in at t=2, as direct action land reform (DALR) participants contest one of the holdings, thereby precipitating CLC. Property control then passes back and forth between DALR participants and the initial occupant, creating a series of management intervals (e.g., t=4 → t=5, t=5 → t=6, etc.). The property without contention experiences ALC until 2,000 ha are cleared, or half the holding, in accordance with federal law that mandates a “Legal Reserve” of 50 percent. The property in contention shows more LC once DALR has initiated (at t=2), and a higher amount of long-run, end-of-period deforestation (at t=10).
We conducted a series of regression analyses, including OLS and fixed-effects in a panel environment, where each property (n = 180) has complete observations for each year (n = 27). The panel data, described above, were integrated at the property level using a digital cadastral map of 180 large landholdings created from the BNP paper map.
The main hypotheses and results:
(H1) land conflict augments deforestation magnitudes,such that:
(H1a) deforestation is greater by the end of study period on properties that have had more stability in control. SUPPORTED. Property stability as the years with only largeholder control increase, 80.57 ha more forest per year
(H1b) property expropriation and settlement formation increases deforestation, reflecting CLC, compared to after when ALC is the dominant driver; SUPPORTED. By the end of the decadal analysis period, properties with CLC present 25.76 ha more deforestation per conflict event
(H2) that deforestation rates are greater during periods of conflict; and SUPPORTED. All independent variables affect deforestation, and deforestation rates are significantly .
(H3) expropriated properties with agrarian reform settlements experience less deforestation than would otherwise be the case given more certain control by smallholder land managers. SUPPORTED.Expropriation and settlement formation increase deforestation, with 58.95 ha more deforestation each year after expropriation occurs.
Last Modified: 04/12/2017 Modified by: Cynthia S Simmons

Please report errors in award information by writing to: [email protected] .

Case Study: Losing the Rainforest, One Tree at a Time
Studying change over time.

Everything changes over time — including people! You might not see a change in yourself from one day to the next, but if you look at a picture of yourself as a baby you can see how much you have changed. Earth's surface changes, too. Some changes are small (a boulder rolls down a hill), or happen quickly (lava flows down the side of a volcano), and we can see a difference from one day to the next. Other changes are too large or too slow to see right away, so we take pictures over time and compare the present to the past, just like you looking at your baby pictures. Information, including pictures, that is collected over time is called "time series" information.
In this chapter, Synthetic Aperture Radar (SAR) satellite images are used to study change in the Amazon rainforest. There are three great reasons for using SAR images to look at the rainforest. First, the rainforest is a cloudy place! Regular cameras cannot take a picture through clouds, but SAR can see right through those clouds – clouds are invisible to SAR. Second, the rainforest is huge! If you were to visit the rainforest in person, it would seem endless. We need to study the "big picture" to tell what changes have occurred, and satellite images give us the biggest picture of all. Third, satellite images are taken of exactly the same places each time the satellite circles Earth, so images from different years match up. You will be able to compare SAR images of the exact same area from three different years to measure how much forest area was lost from a part of the Amazon rainforest between 1994 and 1996. Consider this: Using this technique measures how much forest was lost in just three years. People have been cutting rainforest trees for a long time, with no sign of stopping. What do you think that means for the forest?
Deforesting the Rainforest
Why should anyone care about the Amazon rainforest in Brazil, anyway? Brazil is a long way away, for sure. But today the Amazon River is the source of 20% of all fresh water on Earth, and the Amazon rainforest contains one-third of Earth's remaining rainforests. About 60% of the entire Amazonian rainforest is located in Brazil, and that rainforest affects everybody on our planet — including you!

Almost all living organisms, including you and me, breathe air and need oxygen. The rainforest plants produce about 20% of Earth's oxygen, and absorb almost 20% of the carbon dioxide we release annually by burning fossil fuels 1 . The Amazon is home to some incredible animals, including jaguars, poison dart frogs, scarlet macaws, howler monkeys, and millions of other species, including an estimated 30 million species of insects! Maybe you don't like bugs, but what about monkeys? Beautiful parrots? Big cats, or cool frogs?
Do you like tasty food? Many spices we use every day to make our food taste better come from the rainforest, including vanilla, coconut, cinnamon, and pepper. Chocolate comes from the rainforest, too! Let's talk about medicine. Have you or anyone you cared about ever been sick? Gone to the dentist? Everything from quinine, a medicine for malaria, to vincristine, a potent (really strong) cancer medicine, to novocaine, a local anesthetic (numbing agent) that your dentist uses, and lots more medicines come from rainforest plants. Only a small number of rainforest plants have been tested for medicinal uses.
Question: Who knows how many special plants we lose every year because the rainforests are being cut down? Answer: No one knows. No one at all.
Brazil is not just big trees, lots of bugs, and wild animals. People live there, too! People that cut down rainforest trees are not just inconsiderate or destructive. They want the land for roads and mining and cattle ranches. They sell valuable hardwood trees, like teak, to other countries, including the United States. Poor farmers clear the land so they can grow vegetables to feed their families. People who are pro-development conflict with people, including indigenous (native) people, who want very much to save the rainforest. This is not a simple problem. To learn more about the Amazon rainforest, a great place to start is Raintree .
Forests and Climate Change

In their natural state, forests are a sink, or storehouse, of carbon dioxide (CO 2 ). In other words, they store more CO 2 than they release. According to Dr Pep Canadell, from the Global Carbon Project and CSIRO Marine and Atmospheric Research, deforestation accounts for approximately 20% of global emissions. This is more than the amount released by 13 years of fossil fuel burning.
Scientists estimate that 5.9 gigatons of CO 2 enters the atmosphere every year from deforestation. That destruction amounts to 50 million acres — or an area the size of England, Wales and Scotland felled annually. When tropical forests are cut–down and burned they release the CO 2 that they had stored back to the atmosphere. Therefore, protecting tropical rainforests is not only important to protect biodiversity it is an important tool in our efforts to mitigate climate change.
In this chapter, you obtain SAR satellite images of an area in the rainforest of northwest Brazil where deforestation has occurred and roads have been built. It is easy to see the roads — they look like fish bones in the satellite images. After exploring the images, use a scientific analysis software package, ImageJ, to analyze them and determine how much forest has been lost from this rainforest area in just three years.
« Previous Page Next Page »
- Shrinking Forest - Growing Problem
- Teaching Notes
- Step-by-Step Instructions
- Tools and Data
- Going Further

- About this Site
- Accessibility
Citing and Terms of Use
Material on this page is offered under a Creative Commons license unless otherwise noted below.
Show terms of use for text on this page »
Show terms of use for media on this page »

- None found in this page
- Initial Publication Date: February 10, 2010
- Short URL: https://serc.carleton.edu/39835 What's This?
- Publications
- Conferences & Events
- Professional Learning
- Science Standards
- Awards & Competitions
- Instructional Materials
- Free Resources
- American Rescue Plan
- For Preservice Teachers
- NCCSTS Case Collection
- Science and STEM Education Jobs
- Interactive eBooks+
- Digital Catalog
- Regional Product Representatives
- e-Newsletters
- Bestselling Books
- Latest Books
- Popular Book Series
- Prospective Authors
- Web Seminars
- Exhibits & Sponsorship
- Conference Reviewers
- National Conference • Denver 24
- Leaders Institute 2024
- National Conference • New Orleans 24
- Submit a Proposal
- Latest Resources
- Professional Learning Units & Courses
- For Districts
- Online Course Providers
- Schools & Districts
- College Professors & Students
- The Standards
- Teachers and Admin
- eCYBERMISSION
- Toshiba/NSTA ExploraVision
- Junior Science & Humanities Symposium
- Teaching Awards
- Climate Change
- Earth & Space Science
- New Science Teachers
- Early Childhood
- Middle School
- High School
- Postsecondary
- Informal Education
- Journal Articles
- Lesson Plans
- e-newsletters
- Science & Children
- Science Scope
- The Science Teacher
- Journal of College Sci. Teaching
- Connected Science Learning
- NSTA Reports
- Next-Gen Navigator
- Science Update
- Teacher Tip Tuesday
- Trans. Sci. Learning
MyNSTA Community
- My Collections

The Deforestation of the Amazon
A Case Study in Understanding Ecosystems and Their Value
By Philip Camill
Share Start a Discussion

In this case study, students examine tropical deforestation in the Amazon from the perspective of three dominant stakeholders in the region: a peasant farmer, logger, and environmentalist. As part of the exercise, students perform a cost-benefit analysis of clearing a plot of tropical forest in the Amazon from the perspective of one of these stakeholder groups. Developed for a course in global change biology, this case could also be used in courses in general ecology, environmental science, environmental ethics, environmental policy, and environmental/ecological economics.
Download Case
Date Posted
- Understand the political, cultural, and economic history leading to tropical deforestation in Amazonia. Understand issues facing the major stakeholders in the Amazon.
- Understand the concern for such a large loss in biodiversity.
- Understand the concepts of market and non-market valuation of ecosystems, benefit-cost analysis, and opportunity cost.
- Perform a cost-benefit analysis of clearing a plot of tropical forest in the Amazon, from the perspective of a peasant farmer, logger, and environmentalist.
- Critically evaluate economic vs. ethical valuation of ecosystems.
- Appreciate the political, social, economic, and ecological complexity of tropical deforestation.
- Appreciate how difficult decisions must me made in the face of limited or nonexistent data.
Deforestation; Amazon; tropical forest; rainforest; ecosystem; biodiversity; bioprospecting; ecotourism; ecological economics; cost-benefit analysis; tropics; developing world; South America
Subject Headings
EDUCATIONAL LEVEL
High school, Undergraduate lower division, Undergraduate upper division
TOPICAL AREAS
Ethics, Policy issues, Social issues, Social justice issues
TYPE/METHODS
Teaching Notes & Answer Key
Teaching notes.
Case teaching notes are protected and access to them is limited to paid subscribed instructors. To become a paid subscriber, purchase a subscription here .
Teaching notes are intended to help teachers select and adopt a case. They typically include a summary of the case, teaching objectives, information about the intended audience, details about how the case may be taught, and a list of references and resources.
Download Notes
Answer Keys are protected and access to them is limited to paid subscribed instructors. To become a paid subscriber, purchase a subscription here .
Download Answer Key
Materials & Media
Supplemental materials, you may also like.
Web Seminar
Join us on Thursday, August 15, 2024, from 7:00 PM to 8:00 PM ET, to learn about space weather from NASA. In this web seminar, we will delve into the...
Join us on Wednesday, October 9, 2024, from 7:00 PM to 8:15 PM ET, to learn about NGSS implementation efforts over the past ten years....
Join us on Wednesday, October 2, 2024, from 7:00 PM to 8:15 PM ET, to learn the process that NSTA follows to review conference proposals. Durin...
Join us on Tuesday, August 27, 2024, from 7:00 PM to 8:15 PM ET, to learn about how to submit a successful proposal to present at an NSTA National Con...
- Corpus ID: 146632308
The Deforestation of the Amazon : A Case Study in Understanding Ecosystems and Their Value
- P. Camill , R. Althaus , +3 authors P. Soranno
- Published 1999
- Environmental Science
- Science and Engineering Ethics
3 Citations
Exploring the impact of gamified role-playing on climate change knowledge and nature relatedness: evidence from an online undergraduate course on environmental health, tropical deforestation: can property rights stem the tide, earthworms as a bioindicator of mercury pollution in an artisanal gold mining community, cachoeira do piriá, brazil, related papers.
Showing 1 through 3 of 0 Related Papers
Large differences between observed and expected Ecuadorian deforestation from 2001 to 2009: a counterfactual simulation approach
- Original Article
- Open access
- Published: 01 June 2024
- Volume 24 , article number 94 , ( 2024 )
Cite this article
You have full access to this open access article

- Baltazar Calvas 1 , 2 ,
- Luz Maria Castro 1 , 3 ,
- Mengistie Kindu 1 ,
- Logan Bingham 1 ,
- Karla Pintado 1 ,
- Jonathan Torres Celi 1 , 3 ,
- Thomas Knoke 1 &
- Jorge Cueva-Ortiz ORCID: orcid.org/0000-0002-7920-8343 1
414 Accesses
12 Altmetric
Explore all metrics
Ensuring the integrity of the world’s forests is indispensable for mitigating climate change, combatting biodiversity loss, and protecting the livelihoods of rural communities. While many strategies have been developed to address deforestation across different geographic scales, measuring their impact against a fluctuating background of market-driven forest loss is notoriously challenging. In this article, we (1) asses deforestation in Ecuador using a dynamic, counterfactual baseline that excludes non-market factors, (2) identify periods of reduced and excess deforestation, and (3) assess the economic consequences of associated CO 2 emissions using the social cost of carbon metric. We construct a counterfactual market-forces-only reference scenario by simulating heterogeneous profit-seeking agents making satisficing land-use allocation decisions under uncertainty. The model simulates a reference scenario for 2001–2022, a period encompassing dollarization, the beginning of a constitution granting inalienable rights to nature, and the launch of the largest payments for ecosystem services program in Ecuador’s history. On this period, total deforestation was approximately 20% lower than expected in a market-forces-only scenario (9540 vs.12,000 km 2 ). The largest deviation occurred in 2001–2009, when observed deforestation was 43.6% lower than expected (3720 vs 6590 km 2 ). From 2010 onwards, deforestation appears to be market-driven. We assess the economic value of avoided CO 2 emissions at US $5.7 billion if the reduction is permanent, or US $3.1 billion considering a 1% risk of loss from 2022 onwards. We discuss contributing factors that likely shaped periods of reduced and excess deforestation and stress the need to use realistic baselines.
Similar content being viewed by others

Strategies to achieve a carbon neutral society: a review
The costs and benefits of environmental sustainability
Cork oak woodlands and decline: a social-ecological review and future transdisciplinary approaches
Avoid common mistakes on your manuscript.
Introduction
Global forest area has decreased by roughly 10% in the last 60 years, with the most serious losses occurring in the tropics (Hansen et al. 2013 ; Estoque et al. 2022 ). In addition to their intrinsic value, tropical forests account for more than half of Earth’s terrestrial biodiversity (FAO 2022 ), provide local ecosystem services to approximately 1.5 billion people (Lewis et al. 2015 ), and generate global benefits like carbon sequestration and climate stabilization (Fuss et al. 2021 ). Unfortunately, gross carbon loss from tropical forests is accelerating, with a doubling from 2001 to 2019 that appears to be driven mainly by agricultural expansion (Feng et al. 2022 ). South America is a leading contributor (Hansen et al. 2013 ; Feng et al. 2022 ).
The integrity of the world’s forests, tropical and otherwise, depends heavily on how we interact with and use them (Díaz et al. 2019 ; Purvis et al. 2019 ; IPCC 2022 ). In recent decades, various initiatives operating at international, regional, and local levels have been developed to combat deforestation and mitigate its impacts, such as the UN-REDD+ Program, LEAF Coalition, Zero-Deforestation agreements, and Ecuador’s Socio Bosque program (de Koning et al. 2011 ; Un-REDD programme 2016 ; Pasiecznik and Savenije 2017 ; LEAF Coalition 2023 ). However, these schemes often lack explicit criteria for monitoring and assessment, making it difficult to evaluate their effectiveness (Garrett et al. 2019 ). Even when systematic assessments are employed, such as the Monitoring, Reporting, and Verification (MRV) system used by REDD+ projects, they are typically based on static before-after comparisons of carbon stocks and emissions (IPCC 2022 ), an approach which neglects existing trends and shifting drivers of forest loss. Although it is well-established that using inappropriate baselines can significantly overestimate emissions reductions (West et al. 2020 ; West et al. 2023 ), defining sound “business-as-usual” baselines can be immensely challenging, especially on forest-relevant timescales. As a result, most assessments lack plausible counterfactual reference scenarios that clearly describe what would have happened in the absence of an intervention (Köthke et al. 2014 ; Bos et al. 2017 ; Gifford 2020 ).
Ecuador stands out as one of the world’s most fascinating case studies in forest and biodiversity protection (Coral et al. 2021 ). Situated at the biogeographic confluence of the Andes, the Amazon basin, and the Tumbes-Chocó-Magdalena hotspot (Iturralde‐Pólit et al. 2017 ), Ecuador is a megadiverse country (Kleemann et al. 2022 ) that for decades recorded the highest deforestation rates in South America, losing more than half of its forest area since the 1970s (Bilsborrow et al. 2004 ; Tapia-Armijos et al. 2015 ). Figure 1 compares the forest conditions in 1990 and in 2022 based on public data from the Ministerio del Ambiente, Agua y Transición Ecológica ( 2024 ). The figure illustrates a substantial reduction in the extent of natural forest cover over the 32-year period, as well as the spatial distribution of the forest loss. Deforestation hotspots are concentrated in the Ecuadorian Amazon basin, particularly along the so-called Troncal Amazónica , a major road that crosses the region from north to south. Additionally, two other affected areas were identified along the northern and southern borders in this region. Intense deforestation was also observed in the northern coastal region, while the remaining forests in the central and southern coastal areas, as well as in the western Andean mountain range, experience comparatively lower levels of deforestation. The country has a disproportionately high share of species on the IUCN red list (Rodrigues et al. 2014 ). Although logging, mining, and oil concessions all make significant contributions (Ojeda Luna et al. 2020 ; Sierra et al. 2021 ), agricultural expansion (pasture and croplands) remains the primary driver of deforestation and biodiversity loss in Ecuador (Mena et al. 2011 ; Knoke et al. 2016 ).
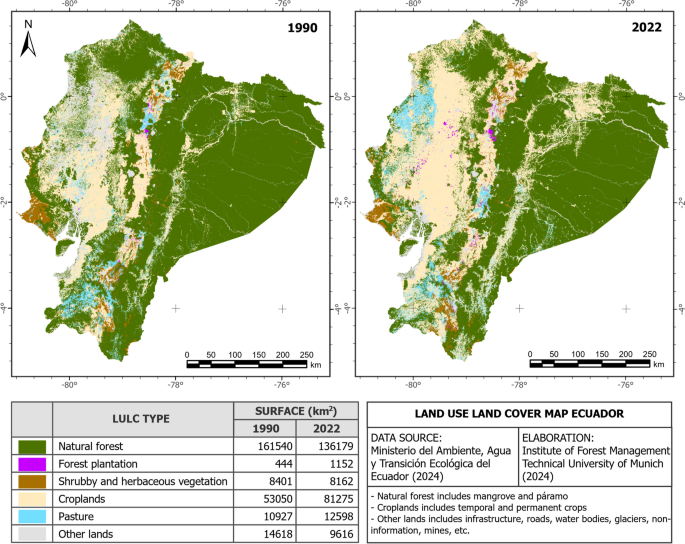
Land-use land-cover (LULC) map of Ecuador illustrating the status of natural forest in 1990 and 2022. Based on data from Environmental, Water and Ecological Transition Ecuadorian Ministry (Ministerio del Ambiente, Agua y Transición Ecológica 2024 )
Following overlapping periods of economic upheaval and nearly a decade (1997–2008) of decentralized natural resource management reform which occurred mainly at the canton (a spatial administrative unit similar to a county) level (Kauffman and Terry 2016 ), in 2008, Ecuador became the first country in the world to constitutionally guarantee specific, legally enforceable rights to nature (Asamblea constituyente del Ecuador 2008 ; Tanasescu 2013 ; Kotzé and Villavicencio Calzadilla 2017 ). These include the right to exist and persist (Article 71), to restoration (Article 72), and to provide environmental benefits to people and communities (Article 74) (Kotzé and Villavicencio Calzadilla 2017 ; Nieto Sanabria 2017 ).
Empowered by this last provision in particular, Ecuador’s Environmental Ministry swiftly implemented a program, Socio Bosque, with the dual goals of conservation and poverty alleviation (de Koning et al. 2011 ; Nieto Sanabria 2017 ). This program offers economic incentives to private owners and communities who willingly pledge to conserve and restore forest and páramo for at least 20 years (Vanacker et al. 2018 ). As of May 2023, nearly 17,000 km 2 have been enrolled, representing more than 6% of continental Ecuador’s total land area, and payments have been disbursed to some 120,000 beneficiaries (Ministerio del Ambiente, Agua y Transición Ecológica 2023 ).
Taken together, it is a remarkable story. A nation that suffered from an economic crisis and was unable to halt the decline of its natural forests decided to undertake a bold legal experiment to constitutionally recognize the rights of nature. The country succeeded in implementing it and immediately launched the largest-ever payments for ecosystem services program in its history. Despite garnering well-deserved attention from scholars, policymakers, and the media, relatively few efforts have been made to systematically quantify the aggregate impact of these changes on national-scale deforestation trends (but see Mohebalian and Aguilar 2016 ; Eguiguren et al. 2019 ; Fischer et al. 2021 ).
In this study, we apply a new method to assess deforestation trends in Ecuador against a dynamic counterfactual baseline, and assess the economic value associated with periods of excess (i.e. above-baseline) and reduced (below-baseline) deforestation using published estimates of the social cost of carbon (SCC). The baseline scenario is designed to reflect the level of deforestation attributable to market forces only (i.e. what would have occurred in a counterfactual scenario where external factors, such as policy interventions, are absent). Deviations between the market-forces-only baseline and observed deforestation levels are assumed to result from these external factors. We investigate the following questions:
Q1 Does the observed deforestation deviate from the market-oriented deforestation trajectory?
Q2 Are the deviations between observed and counterfactual deforestation associated with non-market factors at the country scale?
Q3 What is the economic value of CO 2 emissions associated with excess or reduced deforestation relative to the counterfactual market-forces-only baseline?
To construct a dynamic, counterfactual baseline, we adopt an approach recently introduced by Knoke et al. ( 2023 ) consisting of a behaviourally consistent land-use allocation model in which profit-seeking agents make satisficing decisions under future uncertainty. This approach is based on three assumptions: (a) forest loss is driven by the transformation of land use, mainly into agriculture; (b) decision-makers seek satisfactory and sufficient (“satisficing”), rather than maximal, profits; and (c) the individual expectations of land managers drive land-use allocation decisions. Building on heterogenous expectations of land managers about market prices, agricultural productivity, and corresponding profits, this approach establishes a suitable deforestation baseline for estimating the social value of excess and avoided deforestation.
Material and methods
We compare observed forest cover losses for Ecuador (2000–2022) from Global Forest Watch ( 2024 ) with simulated forest losses from a market-oriented counterfactual land-use allocation model. Because Ecuador’s forest cover is dominated by natural forest, we interpret any forest loss as a permanent removal, neglecting that some forests might re-establish naturally or be replanted on previously cleared lands (see Bos et al. 2017 for a similar assumption and Feng et al. 2022 for empirical support).
Background and expected deforestation trends
Between 1990 and 2000, Ecuador experienced a series of interlinked economic crises (Beckerman 2001 ) and political instability (Conaghan and La Torre 2008 ; Kauffman and Terry 2016 ), culminating in a strong currency depreciation and the adoption of the US dollar as the national currency (“dollarization”). Because currency depreciations and political instability tend to enhance deforestation in developing countries (Didia 1997 ; Arcand et al. 2008 ), it is reasonable to assume that this period featured higher deforestation levels than would expected from purely microeconomic land-use allocation decisions. Conversely, however, this period of national political instability created space for a series of local, canton-level natural resource management reforms, such as measures to combat deforestation in rural watersheds, which were pursued from 1997 to 2008. Kauffman and Terry ( 2016 ) showed that these reforms were attempted by 94 of Ecuador’s 221 cantons, collectively representing about 50% of the country’s area (see Fig. 1 in Kauffman and Terry 2016 ). Combined with the currency stabilization from 2000 onwards, the reforms may have caused reduced deforestation.
In 2006, the election of president Rafael Correa marked a turning point in Ecuador’s political landscape (Conaghan and La Torre 2008 ). Among other things, it opened the door to ratifying a new constitution granting inalienable rights to nature and establishing the Socio Bosque conservation program, both in 2008 (Krause and Loft 2013 ). Intuitively, one would expect these developments to exert a downward pressure on deforestation rates. However, the 2007–2020 period featured a novel combination of large-scale conservation incentives alongside an expansion of “neo-extractivist” activities (Coral et al. 2021 ), opposing trends that may have counteracted one another.
Counterfactual land-use allocation to model expected deforestation
We adopted an approach to counterfactual land-use allocation modelling recently introduced by Knoke et al. ( 2023 ), which aims to support evaluations of broad country-scale non-market drivers of deforestation by simulating plausible reference scenarios corresponding to deforestation levels that would be expected in the absence of any policy intervention (i.e. if land-use allocation decisions were driven exclusively by microeconomic factors). The broad country-level counterfactual deforestation simulation is non-spatial. We simulate land-use allocation decisions (including forest conversion to alternative LULC types) for hypothetical regions of Ecuador, assuming groups of agents with heterogeneous expectations concerning economic profits and uncertainties. These simulations are likely to be really independent from observed deforestation and would thus be well-suitable as reference scenarios. We start with the premise that land-use allocation processes at the regional level, even when agents act individually, are often influenced by suggestions and advice from others. Salas-Molina et al. ( 2023 ) have recently described such simulation approaches in general. Our model is parsimonious in that it uses only input information on agricultural productivity and market prices and costs. The possible variation of the expectations of different land managers is simulated based on random Monte Carlo simulations, assuming that individual land managers perceive future productivities and profits differently. The random process assigns more or less pessimistic profit expectations to more or less risk-averse land managers, so that decision-makers’ heterogeneity is represented. The core principle of our simulation uses a distance function expressing the current economic dissatisfaction of the individual members of a group of agents. We minimize this function across multiple heterogeneous expectations concerning the current and future land-use profits by simulating an appropriate land-use re-allocation.
To address such decision processes, including multiple decision-making agents at the regional scale, we generated counterfactual reference scenarios by simulating a large number of land-use allocation decisions made by heterogeneous groups of agents responding to microeconomic signals. The difference between the empirical (actually observed) deforestation levels and the reference (i.e. simulated counterfactual baseline) represents reduced or excess deforestation, which we use as a basis for calculating the economic value of both avoided and additional carbon emissions, using published estimates for SCC (see Supplementary Methods 4.2 and Supplementary Table 8 in Knoke et al. 2023 ). We used an emission factor of 108 Mg C hectare −1 (derived from data in Harris et al. 2012 ).
In this application, agents represent land manager groups making satisficing decisions under uncertainty in response to subjective profit expectations. Uncertainty encompasses volatility of market prices, productivity fluctuations, and potential crop losses caused by natural hazards and sudden political changes. In our model, Ecuadorian land managers would consider multiple possible future profits. Our approach implies satisficing decisions, which means that land managers gradually improve their current land-use profits by seeking a compromise land-use allocation that promises “sufficient” outcome levels in all possible future states of the world. To achieve this, an individual land manager would consider two types of information: (1) expected profits under the current land-use allocation and (2) the best achievable profits expected under a different allocation (for multiple possible future profit scenarios). The potential for improvement is represented by the difference between (2) and (1), which shows the degree of land manager dissatisfaction with current profits. In contrast to a previous non-stochastic model (Knoke et al. 2020 ), we use a wide range of stochastic profit scenarios to account for the heterogeneity of individual expectations (Grêt-Regamey et al. 2019 ). This implies that not all land managers would contribute equally to LULC changes, but mainly those who perceive the greatest potential for improving their profits (i.e. those who experience the largest dissatisfaction with their current profits). Individual land managers are clustered into groups to represent potential regional heterogeneity, and we assume that such groups arrange their land-use allocation to minimize their maximum dissatisfaction across multiple future profit expectations over time, considering their heterogeneous profit expectations. Individual land managers’ subjective economic expectations are represented as profit scenarios \(p\) , consisting of a profit vector for seven LULC types generated using Monte Carlo simulation (i.e. random draws from regionally simulated profit probability distributions).
where \({R}_{\dots . p}\) represents the random profits of a specific LULC type under profit scenario \(p\) in a given period, \(E\left({R}_{\dots .}\right)\) denotes the mathematical expectation value of the profit expressed in US$ per hectare, the variable \(u\) is a multiplier to obtain the upper interval limit ( \(u=1\) ), while \(sd\) represents the standard deviation of the profits, \({r}_{\#}\) a uniformly distributed random number ( \(0\le {r}_{\#}\le 1\) ), and \(l\) acts as second multiplier to obtain lower interval limit ( \(l=2)\) .
The standard deviations \(sd\) were derived empirically as root means square errors (RMSE) of regression trend lines, parameterized using FAO data on gross production values. The decisions made by landowner groups are satisficing because they do not aim to maximize profits under expected conditions but instead seek a satisfactory and sufficient land-use allocation even under worst-case conditions across multiple future expectations. Satisficing allocations are identified using a modified version of the robust optimization approach described by Knoke et al. ( 2020 ), adjusted to incorporate heterogeneous and stochastic profit expectations. Each land manager group’s objective function minimizes a measure we call dissatisfaction \({D}_{p}\) , defined as the difference between the highest potential profit \({R}_{p}^{*}\) ($ ha −1 ) under assumed best-case conditions in one specific profit scenario \(p\) , and the assumed actually achievable profit obtained through a given future LULC composition \({R}_{p}\) , being \({R}_{p*}\) the lowest individually assumed profit ($ ha −1 )
\({R}_{p}\) is obtained as follows,
with \({N}_{Ti}+{Df}_{Ti}+{P}_{Ti}+{C}_{Ti}+{H}_{Ti}+{S}_{Ti}+{O}_{Ti}=1\) (negative area proportions excluded)
where \({N}_{Ti}{R}_{Np}\) represents the future proportion of natural forest multiplied by the profits of natural forest in profit scenario \(p\) , subject to the condition \({0\le N}_{Ti}\le N\) , where \(N\) denotes the current proportion of natural forest. \({Df}_{Ti}{R}_{Dp}\) describes the future proportion of deforested areas (forest conversion to agriculture) multiplied by the profits of deforestation in profit scenario \(p\) ( \({0\le Df}_{Ti}\le N\) ). \({P}_{Ti}{R}_{Pp}\) is the future proportion of pasture multiplied by the profits of pasture in profit scenario \(p\) , \({C}_{Ti}{R}_{Cp}\) is the future proportion of cropland (annual crops) times profits of cropland in profit scenario \(p\) , \({H}_{Ti}{R}_{Hp}\) stands for the future proportion of permanent crops times profits of permanent crops in profit scenario \(p\) , \({S}_{Ti}{R}_{Sp}\) denotes the future proportion of forest plantations multiplied by the profits of forest plantations in profit scenario \(p\) , and \({O}_{Ti}{R}_{Op}\) signifies the future proportion of other land times the profits of other land (profits are assumed to be zero for other land-use types).
Because each land manager-member in a group is assigned several random profit scenarios, their dissatisfaction with a given land allocation also varies. The simulation-optimization model selects a target land-use portfolio that minimizes the maximum dissatisfaction \({D}_{p}\) for the group as a whole. The current land-use allocation is then incrementally adjusted over the course of a 30-year time horizon to approach the target portfolio. Agents consider allocations across seven LULC types, including natural (i.e. non-plantation) forest \({N}_{Ti}\) . Conversions away from natural forests \({N}_{Ti}\) are counted as forest loss \({L}_{i}\) . To obtain overall reference deforestation levels \({L}_{t}\) , we average \({L}_{i}\) across all 50 groups for each year of the simulation.
where \({L}_{i}\) represents the area of natural forest lost for land manager group \(i\) (ha year −1 ), \(A\) stands for the total land area of a country (ha), and \({N}_{Ti}\) signifies the desired future proportion of natural forest after \(T\) years, with the condition \({0\le N}_{Ti}\le N\) , being \(N\) the current proportion of natural forest (with \(0\le N\le 1\) ). \(k\) represents a factor that takes into account for the planning horizon ( \(k=1\div T\) ), and \(T\) is the planning horizon, assumed to be 30 years ( \(T=30\) ).
For each year of the study period, we simulated decisions by \(n=50\) land manager groups, each consisting of individual members, who are differentiated from one another by the profit scenarios they have been assigned.
After calibrating the model using historical FAO deforestation data for 1990–2000 (FAO Statistics 2022 ), we compared model predictions with empirical forest loss data for the study period to identify deviations between observed deforestation levels and the reference levels that would be expected to arise from microeconomic decision-making alone (see Knoke et al. 2023 and their supplementary information for more details).
As the calibration period coincided with a period of pronounced political and currency instability in Ecuador, our model was calibrated conservatively. Tables 1 and 3 provide the upper and lower ranges of profit expectations (numbers with standard deviation as units) and the coefficients of variation used for deforestation and natural forest areas that we obtained as a result of calibration.
Economic valuation using the social cost of carbon
In the final step, we analyzed deviations between observed and expected deforestation as additional or avoided carbon emissions, which we valued economically using published SCC estimates. Following Groom and Venmans ( 2023 ), we define SCC as the present value of the social cost of climate damages associated with an additional tonne of emitted CO 2 :
where \({D}_{t}\) is the temperature-dependent marginal damage in USD associated with an additional unit of CO 2 emitted and \(r\) is a constant discount rate. We adopted conservative \({SCC}_{t}\) estimates from the United States Government ( 2021 ) for \(r=0.03\) and assume \({D}_{t}\) to increase at a constant annual rate of \(0.0202\) (United States Government 2021 ); see Knoke et al. ( 2023 ) and their supplementary material for details. We calculate the present value of avoided emissions (Groom and Venmans 2023 ) over time horizon T as:
where \({E}_{t}\) represents the social benefit (cost) associated with reduced (excess) deforestation, and \({S}_{t}\) represents the aggregate avoided (additional) emissions, assumed to stabilize at the end of T :
We account for the climate benefits associated with delayed (rather than absolutely avoided) emissions by assuming a constant annual probability \(\rho\) (here, 1%) that carbon stocks will revert to their initial values after T :
Although the social value of avoided emissions accrues at the global level, the opportunity costs associated with retaining natural forest rather than converting it to more profitable (typically agricultural) LULC are typically incurred by individual landowners. However, these opportunity costs vary substantially over the study period due to market fluctuations. We calculate their cumulative present value as:
where \({S}_{t}^{\Delta L}\) is the cumulative change in the total forest area and \({O}_{t}\) represent the time-dependent land opportunity costs.
\({\Delta L}_{t}\) represents the deviation in observed deforestation relative to the simulated market-only counterfactual reference level. As with climate benefits, we consider non-permanence by accounting for a constant annual probability that opportunity costs will revert to the reference level after the study period, using the same parameter values as above:
Depending on the period considered, the land opportunity costs ranged between 137 and 153 US$ per hectare per year (Table 2 ).
Data sources
Data for observed forest losses were obtained from Global Forest Watch ( 2024 ) and FAOSTAT (FAO Statistics 2022 ). FAO-documented data were used to represent observed forest losses for the calibration of the counterfactual model (1990–2000), as remotely sensed forest losses were not available for this period. Profit data, used as input in the land-use allocation model, were derived from gross production values published in FAOSTAT and from previously published materials (Table 3 ).
Gross profits were calculated by dividing gross production values (constant 2014–2016 US$) by the area harvested for crops or area of LULC type (milk and meat). For forest LULC types, we used the timber volume harvested per hectare multiplied by the price derived from export value for naturally regenerating forests, or used modelled volumes (Knoke et al. 2014 ) multiplied by timber prices adopted from the Global Forest Products model for planted forests (Buongiorno 2003 ). Variation coefficients, \(vc\) , were adopted to obtain standard deviations of profits, \(sd.\) The variation coefficient was built on the RMSE of fitted trend lines, \(vc=rmse/\overline{R }\cdot\;100.\) The LULC category “Deforestation” had the same coefficients as “Permanent meadows & pastures” but a different \(vc\) (Table 3 ). Following Knoke et al. ( 2023 , supplementary material) we assumed that the upfront profits from clearing the natural forest were high enough to finance the new establishment of new pasture.
Simulated counterfactual deforestation rate
The overall average annual deforestation rate for 1990–2022 predicted by the counterfactual market-oriented land-use allocation model was 0.47%, with a maximum of 0.58% p.a. in the period 2005–2009 (a period with very high crop prices) and a minimum of 0.35% p.a. in the periods 2010–2014 and 2015–2022 (a period of declining crop prices). In this section, we use random 5-year periods to convey overall overview of deforestation (Table 3 ).
The frequency distribution of simulated counterfactual deforestation rates followed a negative exponential distribution (Fig. 2 ) with a median deforestation rate of 0.32% p.a. The average simulated forest loss without the influence of non-market factors is 605 km 2 per annum in the period 1990–2022, including the calibration period (1990–2000). For 2001–2022, we obtained an expected deforestation reference level of 545 km 2 p.a., while the observed deforestation was 434 km 2 p.a.
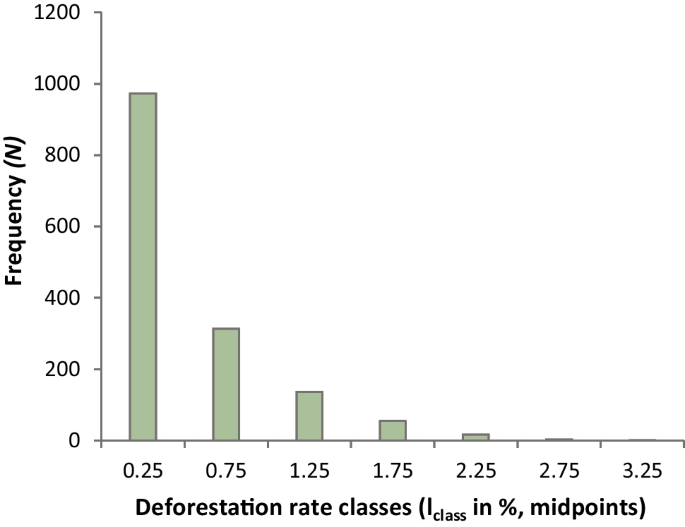
Frequency distribution of simulated deforestation rates by l class . l class is the midpoint of the loss class, shown on the x -axis. The power law behind the distribution of the deforestation rates is described by a trendline with the following coefficients: N = 2090.1 exp (− 2.288 l class ). N is the frequency of the deforestation rates per loss class
Deforestation trends
We calibrate our non-spatial model conservatively to stablish reference deforestation levels slightly below those reported by the FAO (Fig. 3 ). These reference levels capture natural forest losses that would have been expected to occur in the absence of external factors like policy interventions.
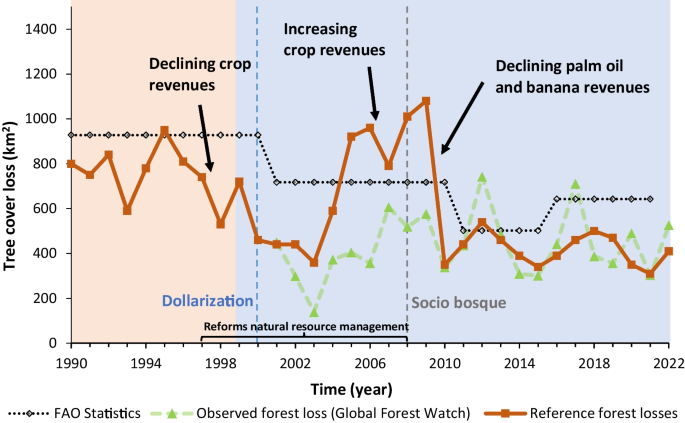
Observed and reference forest cover losses. Observed forest losses refer to tree cover losses (Global Forest Watch 2024 ). Forest losses from the Food and Agriculture Organization of the United Nations (FAO Statistics 2022 ) are compared and were used for calibration (1990–1999) in the absence of remotely sensed data. Light orange shadow represents the calibration period and light blue represents the assessed period. Dashed lines mean the beginning of dollarization (blue) and Socio Bosque program (grey)
The results show large negative deviations between observed and expected counterfactual deforestation for 2001–2009 (Fig. 3 ). This period overlaps with canton-level “reforms of natural resource management” (1997–2008) and starts 1 year after the currency change (“dollarization” in 2000). Our counterfactual market-only scenario thus predicts a trend change of forest losses from 2000 to 2009 in which much less deforestation actually occurred than expected. From 2005 to 2009 in particular, high crop prices led our counterfactual model to predict accelerating deforestation, a trend that did not materialize on the ground. When crop prices declined from 2010 onwards, the observed and expected deforestation do not show systematic deviations, suggesting a period with market-oriented deforestation.
Social value of the deforestation trend
The estimated cumulative avoided emissions from 2000 to 2010 are + 0.114 gigatonnes CO 2 (Fig. 4 a). To calculate the economic value of these avoided emissions, we use conservative SCC ranging from 30.1 (2000) to 44.1 US$ per Mg CO 2 emission (2019) (in 2015 US$ per tonne of CO 2 , suggested by United States Government 2016 ), following the calculations reported in Knoke et al. ( 2023 ). This assessment is based on below-baseline deforestation rates from 2001 to 2009, since observed deforestation closely paralleled the counterfactual baseline from 2010 until the end of the study period.
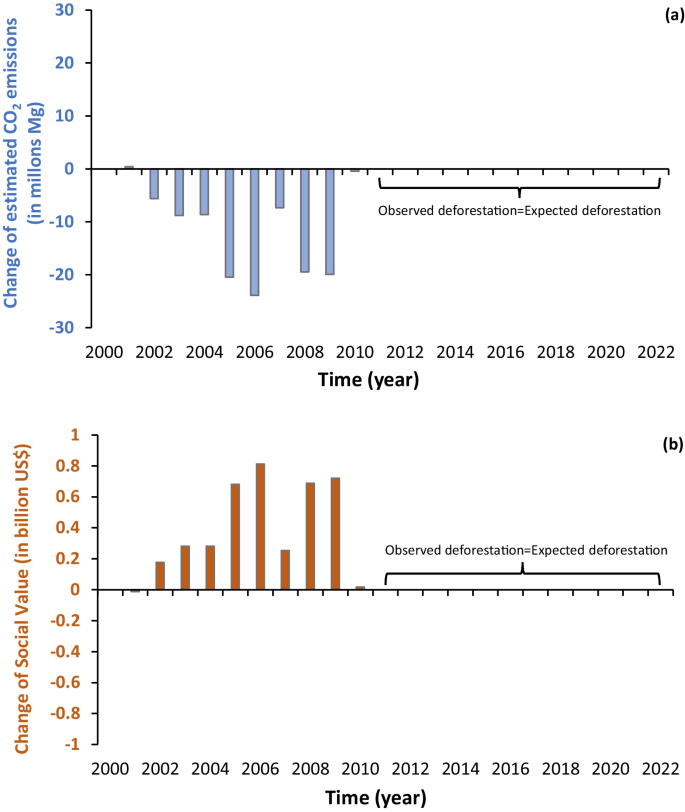
Social values of deforestation in a given year (2000–2009). a Estimated changes in CO 2eq emissions from observed minus counterfactual forest losses. b Associated impact on the social value
The social value of the climate benefits associated with such reduced CO 2 emissions was estimated at $5.7 billion (assuming the achieved emission reductions are permanent, Fig. 4 b) or $3.1 billion (assuming a risk of 1% p.a. of losing the achieved emission reductions after 2022). The foregone agricultural benefits incurred by conserving tropical forests to achieve the climate benefits were estimated at $1.4 billion. Based on these evaluations, we obtain a rough benefit-cost ratio of 2.2.
The comparison of observed deforestation with our simulated market-forces-only baseline shows a substantial conservation of Ecuadorian natural forests of a size of 2872 km 2 between 2001 and 2009. According to our model, if deforestation decisions had been purely market-driven in this period, this forest cover would have been cleared. This is a remarkable result, considering that this period followed one of Ecuador’s major crises in the late 1990s, during which the economy contracted and inflation surged by approximately 60% (Jokisch and Pribilsky 2002 ). Food prices in Ecuador increased substantially from 2000 to 2008 (Gilbert 2010 ), stimulating demand for agricultural land (Harding et al. 2021 ). During this period, our counterfactual simulations predicted accelerating deforestation in response to profit expectations alone, but this expectation was largely unrealized.
One interpretation might hold that the natural resource management reforms pursued by many Ecuadorian cantons between 1997 and 2008, potentially supported by higher currency stability as a consequence of the “dollarization” in 2000, may have been more effective than previously thought (Kauffman and Terry 2016 ).
Conversely, however, some evidence suggests that the dollarization of the national currency at a controversial rate of 25,000 sucres per dollar likely exacerbated already-high emigration rates, which ultimately led to approximately 4% of the population leaving Ecuador (Acosta et al. 2014 ). The period 2000–2004 was particularly difficult for the agricultural sector, as the shuttering of various financial institutions in the years leading up to dollarization created a credit crunch (Chuncho Juca et al. 2021 ). Thus, an alternative explanation might be that the unexpectedly low deforestation rate between 2001 and 2009 was less the result of intentional policy choices or natural resource management reforms than it was a byproduct of inter-connected social, financial, and demographic crises contributing to farm abandonment, rural outmigration, and constraints on the labour resources needed to carry out deforestation.
In January 2007, a new government promising to support conservation came to power (Acosta et al. 2014 ). Its initiatives included, inter alia , the recognition of the rights of nature (Martínez 2021 ), conservation incentives like the Socio Bosque program, and compensation programs for reduced oil exploration and exploitation (Moreano Venegas and Bayón, 2021 ). Insofar as the 2008 constitution presented a groundbreaking legal framework, its implementation has faced persistent challenges from powerful business interests and weak law enforcement (Kauffman and Martin 2017 ). Thus, the effectiveness of Ecuador’s post-2008 environmental policy portfolio is generally viewed as inconsistent.
The Socio Bosque program was launched in 2008 with the aim of conserving forests and native grasslands and improving the livelihoods of rural populations (de Koning et al. 2011 ). Even though the compensation paid to beneficiaries typically falls short of the estimated land opportunity cost, from 2008 to 2017, the Socio Bosque program placed 16,700 km 2 of forest, mangroves, and páramo under protection—more than any other program in the country (Ministerio del Ambiente de Ecuador 2019 ). For comparison, the National Park System ( Sistema Nacional de Areas Protegidas , SNAP) included just 6320 km 2 in the same period, while an additional 3800 km 2 were classified under the Protective Forests and Vegetation ( Bosques y Vegetación Protectores , BVP) category (Ministerio del Ambiente de Ecuador 2019 ). Thus, the Socio Bosque program was expected to strongly decrease deforestation.
Our analysis is at least initially consistent with that expectation. Due to increasing crop revenues, our counterfactual market-forces-only baseline predicts much higher deforestation levels than are observed from 2003 to 2008, a period of expanding natural resource management reform (Fig. 3 ). The divergence between expected and observed deforestation is especially prominent from 2008 to 2010, the first 2 years of the Socio Bosque program, with Ecuador losing 1000 km 2 less natural forest than expected. However, one can argue that this deforestation trend also appears to be partly influenced by the collapse of palm oil prices during 2008 and 2009, which brought serious problems to small producers in Ecuador (Potter 2011 ).
Unfortunately, this reduction in deforestation does not persist: observed and expected deforestation reconverge in 2010 and generally remain in agreement until the end of the study period, apart from two notable deforestation spikes in 2012 and 2017. This post-2010 reconvergence is driven mainly by a sharp decrease in the level of expected deforestation due to declining palm oil and banana revenues, rather than a sustained increase in observed deforestation (Fig. 3 ). Falling palm oil prices at this time placed significant economic pressure on small producers (Potter 2011 ; Castellanos-Navarrete et al. 2021 ), making the prospect of clearing forest for oil palm plantations substantially less attractive (Vijay et al. 2016 ) and reducing the simulated deforestation rate.
Thus, while the Socio Bosque program likely plays an important role in shaping forest cover in Ecuador, national-scale deforestation trends are unavoidably shaped by a confluence of factors whose interacting effects are difficult to disentangle. Our method only enables the identification of aggregate effects which must be critically analyzed alongside supporting evidence concerning to social, economic, and policy developments. The launch of the Socio Bosque program, for instance, coincided with the early stages of an important economic turnaround in Ecuador (The World Bank 2023 ), facilitated in part by a second oil boom (Cueva and Díaz 2022 ), as well as with a series of biofuel initiatives that increased the extent of palm oil plantations across a number of Latin American countries (Furumo and Aide 2017 ). Dynamics of this nature tend to increase the level of deforestation that would be expected in a market-forces-only scenario.
What about the anomalous spikes in observed deforestation that occurred in 2012 and 2017? The first of these spikes roughly coincides with the establishment of two new large-scale hydroelectric projects, the Coca Codo Sinclair Dam and the Sopladora Hydroelectric Power Plant, in 2010 and 2011, respectively (Vallejo et al. 2019 ). The construction of such megastructures typically exacerbates deforestation both directly (i.e. forest displaced by dams, reservoirs, roads, and transmission lines) and indirectly by stimulating activities associated with deforestation while increasing accessibility to remote forest areas (Marques Da Silva et al. 2018 ). Preliminary evidence suggests that Ecuador is unlikely to present a major exception to these well-documented patterns (Finer and Jenkins 2012 ; Vallejo et al. 2019 ; Llerena-Montoya et al. 2021 ).
The 2017 spike, in turn, coincides with the completion of both hydroelectric projects, widespread mortality in Amazon forests associated with a particularly strong El Niño event in 2015–2016 (Berenguer et al. 2021 ), and a legislative effort to loosen regulations regarding mining concessions in protected forests in particular (Roy et al. 2018 ). This deregulation effort directly contributed to a fourfold increase in the area subject to mining exploration (Vandegrift et al. 2018 ). The land category most affected by this regulatory change ( Bosques y Vegetación Protectores , BVP) was first codified the 1980s, and thus was in place for the entirety of our calibration period.
Other relevant factors implemented by the Ecuadorian government during the study period that may have influenced deforestation trends include:
Water funds
Three funds aiming to protect watersheds through integrated water management, FONAG, FONAPA, and FORAGUA, were inaugurated in 2000, 2008, and 2009, respectively. These projects coincided with the period of canton-level natural resource management reform (Kauffman and Terry 2016 ). As of 2019, they incorporated 4614 km 2 of natural forests and private, communal, or acquired lands designated for restoration (Earth Innovation Institute 2019 ).
Command and control policies
The most significant command-and-control anti-deforestation initiative in recent decades was probably the expansion of national park area under the National System of Protected Areas (Utreras et al. 2017 ). Over the course of study period, 33 protected areas were established, covering 8560 km 2 , with most protected areas located in the Amazon region (Ministerio del Ambiente, Agua y Transición Ecológica 2021 ).
Land tenure
Our study period also encompassed the implementation of two land tenure policies. First, the Land Plan (2009–2013) aimed to foster cooperative approach to land management. Second, the Land Adjudication and Mass Legalization Project (2014–2018) focused on acquiring, redistributing, and legalizing state, private, and vacant properties, with a particular emphasis on agricultural productivity (potentially exacerbating deforestation, see Tanner and Ratzke 2022 ).
Overall, our results show that even temporarily effective forest conversation has high social value in terms of avoiding climate-driven damages. The additional forest conservation achieved between 2001 and 2010, worth approximately $3.1 billion, easily outweighs the estimated land opportunity cost of $1.4 billion associated with conservation. The economic benefits of the additional carbon storage alone thus suggest a benefit-to-cost ratio of 2.1. Crucially, the social value of tropical forest is substantially higher than its carbon storage value (e.g. Franklin and Pindyck 2018 ).
Limitations and strengths
Assessing the reliability of our results means evaluating the plausibility of our simulated baseline scenario, the accuracy of observed deforestation data, and assumptions about the future status of newly harvested forest parcels. Because the baseline scenario is designed to capture something that is empirically unobservable—what would have happened under different circumstances—it is difficult to conclusively validate using traditional means, such as experimental controls. Instead, we demonstrate plausibility mainly by showing that the model is able to capture deforestation trends effectively by the model implementation under distinct social conditions and deforestation scales (Fig. 5 ). Although asserting that deforestation in the calibration period was shaped by market forces alone would be a step too far, the overall correlation between observed and simulated deforestation is rather high. The response of deforestation simulations to changes in crop prices (particularly banana and palm oil) is not only facially plausible but also accord well with past empirical work (Taheripour et al. 2019 ; Gaveau et al. 2022 ).
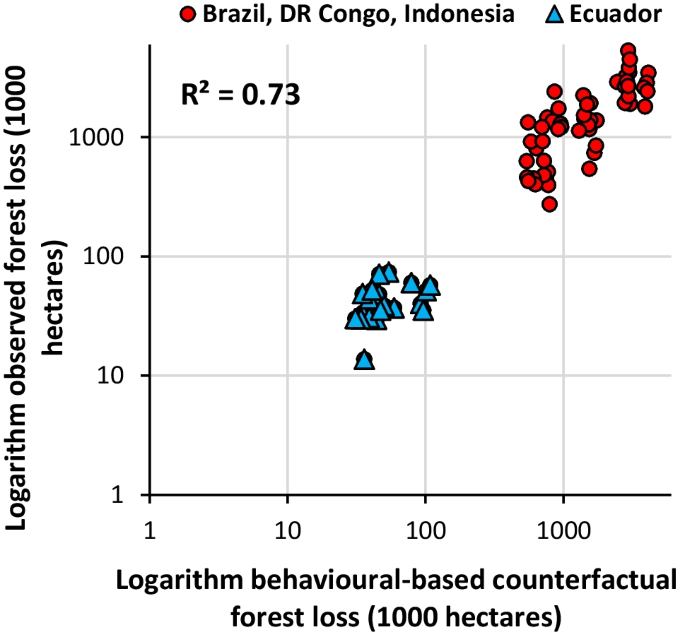
Behaviour of our counterfactual model implemented in countries under different social and political conditions, and thus deforestation scales (data for Brazil, DR Congo, and Indonesia from Knoke et al. 2023 )
Regarding the data used to capture observed deforestation, there was a technical change in the accuracy of the forest loss estimates from Global Forest Watch after 2015 (Weisse and Potapov 2021 ). However, we do not identify a systematic shift of observed deforestation levels when comparing the periods 2010–2014 and 2015–2022 that could systematically bias our results (Fig. 3 ). We note that we used forest loss data specifically, which includes canopy removals that are not necessarily permanent and which might be expected to regenerate. Empirically, however, most recent deforestation in Ecuador is for agriculture, which is rarely followed by a land-use change back to forest (Feng et al. 2022 ). In sum, we expect that our market-oriented reference scenario is plausible and that the observed deforestation levels are valid (because excluding non-market factors rather too high than too low).
Conclusions
Using counterfactual land-use modelling, we estimate the aggregate effect of non-market factors like policy reform and conservation initiatives on national-scale deforestation trajectories in Ecuador. During a study period that encompassed large political, economic, and social change—including the ratification of a constitution granting rights to nature and the introduction of the largest payments for ecosystem services program in Ecuador’s history—we identify substantial reductions in deforestation relative to a market-forces-only counterfactual. This suppressed deforestation appears to have spared some 2872 km 2 of forest—an area roughly equivalent to the Ecuadorian provinces of Santa Elena or Carchi—between 2000 and 2010. Depending on assumptions about the permanence of emissions reductions, this corresponds to a delayed or avoided social cost of carbon in the range of $3.1–$5.7 billion, easily outweighing the value of foregone agricultural production.
While our method only allows for the estimation of aggregate trends and not causal attributions to specific events or policy initiatives, it is plausible that policies such as the Socio Bosque program, which placed more surface area under protection than any other program in Ecuador, likely contributed to decreasing deforestation at least temporarily, even relative to a dynamic baseline. Moving forward, efforts to develop and refine mechanisms that reward land managers for their role in sustaining the high social value that tropical natural forests contribute to global society need further support. In conjunction with efforts to quantify the social value of the ecosystem services provided by tropical forests, research and policy should continue to develop strategies to foster the integration of local stakeholders into tropical land-use policy and decision-making.
Acosta A, Arcos Cabrera C, Ávila Santamaría R, Corral L, Cuvi J et al. (2014) La restauración conservadora del correísmo. Arcoiris Producción Gráfica, Quito Ecuador
Arcand J-L, Guillaumont P, Jeanneney SG (2008) Deforestation and the real exchange rate. J Devel Econ 86(2):242–262. https://doi.org/10.1016/j.jdeveco.2007.02.004
Article Google Scholar
Asamblea Nacional del Ecuador (Ed.), 2008. Constitución de la República del Ecuador, 449th ed., Ecuador
Beckerman P (2001) Dollarization and semi-dollarization in Ecuador. Policy Research Working Paper 2643
Berenguer E, Lennox GD, Ferreira J, Malhi Y, Aragão LEOC, et al. (2021) Tracking the impacts of El Niño drought and fire in human-modified Amazonian forests. Proc Nat Acad Sci United States Am 118(30):e2019377118. https://doi.org/10.1073/pnas.2019377118
Article CAS Google Scholar
Bilsborrow RE, Barbieri AF, Pan W (2004) Changes in population and land use over time in the Ecuadorian Amazon. Acta Amaz. 34(4):635–647. https://doi.org/10.1590/S0044-59672004000400015
Bos AB, Duchelle AE, Angelsen A, Avitabile V, de Sy V, et al. (2017) Comparing methods for assessing the effectiveness of subnational REDD+ initiatives. Environ Res Lett 12(7):74007. https://doi.org/10.1088/1748-9326/aa7032
Buongiorno J (2003) The global forest products model: structure, estimation, and applications. https://buongiorno.russell.wisc.edu/gfpm/ . Accessed 14 May 2022
Castellanos-Navarrete A, de Castro F, Pacheco P (2021) The impact of oil palm on rural livelihoods and tropical forest landscapes in Latin America. J Rural Stud 81:294–304. https://doi.org/10.1016/j.jrurstud.2020.10.047
Chrisendo D, Siregar H, Qaim M (2021) Oil palm and structural transformation of agriculture in Indonesia. Agric Econ 52(5):849–862. https://doi.org/10.1111/agec.12658
Chuncho Juca L, Uriguen Aguirre P, Apolo Vivanco N (2021) Ecuador: análisis económico del desarrollo del sector agropecuario e industrial en el periodo 2000–2018. RCTU-UPSE 8(1):8–17. https://doi.org/10.26423/rctu.v8i1.547
Conaghan C, de La Torre C (2008) The permanent campaign of Rafael Correa: making Ecuador’s plebiscitary presidency. Int J Press/Politics 13(3):267–284. https://doi.org/10.1177/1940161208319464
Coral C, Bokelmann W, Bonatti M, Carcamo R, Sieber S (2021) Understanding institutional change mechanisms for land use: lessons from Ecuador’s history. Land Use Policy 108:105530. https://doi.org/10.1016/j.landusepol.2021.105530
Cueva S, Díaz JP (2022) The History of Ecuador. In: Kehoe TJ, Nicolini JP (eds) A monetary and fiscal history of Latin America. University of Minnesota Press, Minneapolis, pp 1960–2017
Google Scholar
de Koning F, Aguiñaga M, Bravo M, Chiu M, Lascano M, et al. (2011) Bridging the gap between forest conservation and poverty alleviation: the Ecuadorian Socio Bosque program. Environ Sci Policy 14(5):531–542. https://doi.org/10.1016/j.envsci.2011.04.007
Díaz S, Settele J, Brondízio ES, Ngo HT, Agard J, et al. (2019) Pervasive human-driven decline of life on Earth points to the need for transformative change. Science (New York, N.Y.) 366:6471. https://doi.org/10.1126/science.aax3100
Didia DO (1997) Democracy, political instability and tropical deforestation. Global Environ Change 7(1):63–76. https://doi.org/10.1016/S0959-3780(96)00024-6
Earth Innovation Institute (2019) Evaluación del Impacto de políticas públicas destinadas a reducir la deforestación y degradación y acciones destinadas a la gestión sostenible de los bosques en Ecuador: Producto 2. Earth Innovation Istitute. http://proamazonia.org/wp-content/uploads/2020/01/EIIProductoDos-Evaluacio%CC%81n-de-Impacto-Ecuador_P2-min.pdf . Accessed 25 September 2023
Eguiguren P, Fischer R, Günter S (2019) Degradation of ecosystem services and deforestation in landscapes with and without incentive-based forest conservation in the Ecuadorian Amazon. Forests 10(5):442. https://doi.org/10.3390/f10050442
Estoque RC, Dasgupta R, Winkler K, Avitabile V, Johnson BA, et al. (2022) Spatiotemporal pattern of global forest change over the past 60 years and the forest transition theory. Environ Res Lett 17(8):84022. https://doi.org/10.1088/1748-9326/ac7df5
FAO (2022) The state of the world’s forest 2022: forest pathways for green recovery and building inclusive, resilient and sustainable economies. FAO, Rome
FAO Statistics (2022) FAOSTAT. Food and Agriculture Organization of the United Nations. https://www.fao.org/faostat/en/#home . Accessed 8 May 2022
Feltran-Barbieri R, Féres JG (2021) Degraded pastures in Brazil: improving livestock production and forest restoration. Royal Soc Open Sci 8(7):201854. https://doi.org/10.1098/rsos.201854
Feng Y, Zeng Z, Searchinger TD, Ziegler AD, Wu J, et al. (2022) Doubling of annual forest carbon loss over the tropics during the early twenty-first century. Nat Sustain 5(5):444–451. https://doi.org/10.1038/s41893-022-00854-3
Finer M, Jenkins CN (2012) Proliferation of hydroelectric dams in the Andean Amazon and implications for Andes-Amazon connectivity. PloS One 7(4):e35126. https://doi.org/10.1371/journal.pone.0035126
Fischer R, Tamayo Cordero F, Ojeda Luna T, Ferrer Velasco R, DeDecker M, et al. (2021) Interplay of governance elements and their effects on deforestation in tropical landscapes: quantitative insights from Ecuador. World Devel 148:105665. https://doi.org/10.1016/j.worlddev.2021.105665
Franklin SL, Pindyck RS (2018) Tropical forests, tipping points, and the social cost of deforestation. Ecol Econ 153:161–171. https://doi.org/10.1016/j.ecolecon.2018.06.003
Furumo PR, Aide TM (2017) Characterizing commercial oil palm expansion in Latin America: land use change and trade. Environ Res Lett 12(2):24008. https://doi.org/10.1088/1748-9326/aa5892
Fuss S, Golub A, Lubowski R (2021) The economic value of tropical forests in meeting global climate stabilization goals. Glob. Sustain. 4:e1. https://doi.org/10.1017/sus.2020.34
Garrett RD, Levy S, Carlson KM, Gardner TA, Godar J, et al. (2019) Criteria for effective zero-deforestation commitments. Global Environ Change 54:135–147. https://doi.org/10.1016/j.gloenvcha.2018.11.003
Gaveau DLA, Locatelli B, Salim MA, Husnayaen MT, Descals A, et al. (2022) Slowing deforestation in Indonesia follows declining oil palm expansion and lower oil prices. PloS One 17(3):e0266178. https://doi.org/10.1371/journal.pone.0266178
Gifford L (2020) “You can’t value what you can’t measure”: a critical look at forest carbon accounting. Climatic Change 161(2):291–306. https://doi.org/10.1007/s10584-020-02653-1
Gilbert CL (2010) How to understand high food prices. J Agri Econ 61(2):398–425. https://doi.org/10.1111/j.1477-9552.2010.00248.x
Global Forest Watch (2024) Ecuador Deforestation Rates & Statistics | GFW. Global Forest Watch. https://www.globalforestwatch.org/dashboards/country/ECU/?category=forest-change . Accessed 5 March 2024
Grêt-Regamey A, Huber SH, Huber R (2019) Actors’ diversity and the resilience of social-ecological systems to global change. Nat Sustain 2(4):290–297. https://doi.org/10.1038/s41893-019-0236-z
Groom B, Venmans F (2023) The social value of offsets. Nature 619(7971):768–773. https://doi.org/10.1038/s41586-023-06153-x
Hansen MC, Potapov PV, Moore R, Hancher M, Turubanova SA, et al. (2013) High-resolution global maps of 21st-century forest cover change. Science (New York N.Y.) 342(6160):850–853. https://doi.org/10.1126/science.1244693
Harding T, Herzberg J, Kuralbayeva K (2021) Commodity prices and robust environmental regulation: evidence from deforestation in Brazil. J Environ Econ Manage 108:102452. https://doi.org/10.1016/j.jeem.2021.102452
Harris NL, Brown S, Hagen SC, Saatchi SS, Petrova S, et al. (2012) Baseline map of carbon emissions from deforestation in tropical regions. Science (New York, N.Y.) 336(6088):1573–1576. https://doi.org/10.1126/science.1217962
IPCC (2022) Climate change 2022: impacts, adaptation and vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change, Cambridge, UK and New York, NY, USA. https://report.ipcc.ch/ar6/wg2/IPCC_AR6_WGII_FullReport.pdf . Accessed 06.2023
Iturralde-Pólit P, Dangles O, Burneo SF, Meynard CN (2017) The effects of climate change on a mega-diverse country: predicted shifts in mammalian species richness and turnover in continental Ecuador. Biotropica 49(6):821–831. https://doi.org/10.1111/btp.12467
Jokisch B, Pribilsky J (2002) The panic to leave: economic crisis and the “new emigration” from Ecuador. Inter Migrat 40(4):75–102. https://doi.org/10.1111/1468-2435.00206
Kauffman CM, Martin PL (2017) Can rights of nature make development more sustainable? Why some Ecuadorian lawsuits succeed and others fail. World Devel 92:130–142. https://doi.org/10.1016/j.worlddev.2016.11.017
Kauffman CM, Terry W (2016) Pursuing costly reform: The case of Ecuadorian natural resource management. Latin Am Res Rev 51(4):163–185. https://doi.org/10.1353/lar.2016.0054
Kleemann J, Koo H, Hensen I, Mendieta-Leiva G, Kahnt B, et al. (2022) Priorities of action and research for the protection of biodiversity and ecosystem services in continental Ecuador. Biol Conserv 265:109404. https://doi.org/10.1016/j.biocon.2021.109404
Knoke T, Bendix J, Pohle P, Hamer U, Hildebrandt P, et al. (2014) Afforestation or intense pasturing improve the ecological and economic value of abandoned tropical farmlands. Nat Commun 5:5612. https://doi.org/10.1038/ncomms6612
Knoke T, Calvas B, Aguirre N, Román-Cuesta RM, Günter S, et al. (2009) Can tropical farmers reconcile subsistence needs with forest conservation? Front Ecol Environ 7(10):548–554. https://doi.org/10.1890/080131
Knoke T, Hanley N, Roman-Cuesta RM, Groom B, Venmans F, et al. (2023) Trends in tropical forest loss and the social value of emission reductions. Nat Sustain 6:1373–1384. https://doi.org/10.1038/s41893-023-01175-9
Knoke T, Paul C, Hildebrandt P, Calvas B, Castro LM, et al. (2016) Compositional diversity of rehabilitated tropical lands supports multiple ecosystem services and buffers uncertainties. Nat Commun 7:11877. https://doi.org/10.1038/ncomms11877
Knoke T, Paul C, Rammig A, Gosling E, Hildebrandt P, et al. (2020) Accounting for multiple ecosystem services in a simulation of land-use decisions: Does it reduce tropical deforestation? Global Change Biol. https://doi.org/10.1111/gcb.15003
Köthke M, Schröppel B, Elsasser P (2014) National REDD+ reference levels deduced from the global deforestation curve. Forest Policy Econ 43:18–28. https://doi.org/10.1016/j.forpol.2014.03.002
Kotzé LJ, Villavicencio Calzadilla P (2017) Somewhere between rhetoric and reality: environmental constitutionalism and the rights of nature in Ecuador. TEL 6(3):401–433. https://doi.org/10.1017/S2047102517000061
Krause T, Loft L (2013) Benefit distribution and equity in Ecuador’s Socio Bosque Program. Soc Nat Res 26(10):1170–1184. https://doi.org/10.1080/08941920.2013.797529
LEAF Coalition (2023) LEAF Coalition. Emergent. https://leafcoalition.org/ . Accessed 6 June 2023
Lewis SL, Edwards DP, Galbraith D (2015) Increasing human dominance of tropical forests. Science (New York, N.Y.) 349(6250):827–832. https://doi.org/10.1126/science.aaa9932
Llerena-Montoya S, Velastegui-Montoya A, Zhirzhan-Azanza B, Herrera-Matamoros V, Adami M, et al. (2021) Multitemporal analysis of land use and land cover within an oil block in the Ecuadorian Amazon. IJGI 10(3):191. https://doi.org/10.3390/ijgi10030191
Marques Da Silva O, Dos Santos MA, Sousa L (2018) Spatiotemporal patterns of deforestation in response to the building of the Belo Monte hydroelectric plant in the Amazon basin. Interciencia 43(2):80–84
Martínez E (2021) Mucha Investigación, Poca Traducción, in: Moreano Venegas, M., Bayón, M. (Eds.), La explotación del Yasuní. En medio del derrumbe petrolero global, vol. 1. Editorial Abya-Yala, Quito, pp 79–82
Meade B, Puricelli E, McBride W, Valdes C, Hoffman L, Foreman L, Dohlman E (2016) Corn and Soybean production costs and export competitiveness in Argentina, Brazil, and the United States 154. USDA Economic Information Bulletin, pp 52. https://ssrn.com/abstract=2981675
Mena CF, Walsh SJ, Frizzelle BG, Xiaozheng Y, Malanson GP (2011) Land use change on household farms in the Ecuadorian Amazon: design and implementation of an agent-based model. Appl Geography (Sevenoaks, England) 31(1):210–222. https://doi.org/10.1016/j.apgeog.2010.04.005
Ministerio del Ambiente de Ecuador (2019) Proyecto Socio Bosque. https://www.ambiente.gob.ec/wp-content/uploads/downloads/2020/07/12.SOCIO_BOSQUE.pdf . Accessed 26 September 2023
Ministerio del Ambiente, Agua y Transición Ecológica (2021) Reporte Sistema Nacional de Áreas Protegidas: Periodo 2021 (Cifras Oficiales). https://www.ambiente.gob.ec/wp-content/uploads/downloads/2022/03/reporte_comunica_snap_2021.pdf . Accessed 12.2023
Ministerio del Ambiente, Agua y Transición Ecológica (2023) Proyecto Socio Bosque. https://sociobosque.ambiente.gob.ec/ . Accessed 15 May 2023
Ministerio del Ambiente, Agua y Transición Ecológica (2024) Cobertura y Uso de la Tierra: Mapa Interactivo. http://ide.ambiente.gob.ec:8080/mapainteractivo/ . Accessed 03.2024
Mohebalian PM, Aguilar FX (2016) Additionality and design of forest conservation programs: insights from Ecuador’s Socio Bosque Program. Forest Policy Econ 71:103–114. https://doi.org/10.1016/j.forpol.2015.08.002
Moreano Venegas M, Bayón M (eds) (2021) La explotación del Yasuní: En medio del derrumbe petrolero global. Editorial Abya-Yala, Quito, p 180
Nieto Sanabria L (2017) The subalternization of a progressive legal project: the rights of nature in Ecuador. Mex Law Rev 1(20):117. https://doi.org/10.22201/iij.24485306e.2018.20.11895
Ojeda Luna T, Eguiguren P, Günter S, Torres B, Dieter M (2020) What drives household deforestation decisions? Insights from the Ecuadorian lowland rainforests. Forests 11(11):1131. https://doi.org/10.3390/f11111131
Pasiecznik N, Savenije H (eds) (2017) Zero deforestation: a commitment to change. ETFRN, Wageningen, p 227
Potter LP (2011) La Industria Del Aceite De Palma En Ecuador: Un Buen Negocio Para Los Pequeños Agricultores? Eutopia 2. https://doi.org/10.17141/eutopia.2.2010.1028
Purvis A, Molnar Z, Obura D, Ichii K, Willis K, et al. (2019) Chapter 2.2. Status and Trends – Nature. In: Brondízio ES, Settele J, Diaz S, Ngo HT (eds) Global assessment report of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services. IPBES secretariat, Bonn, Germany, pp 202–310
Rodrigues ASL, Brooks TM, Butchart SHM, Chanson J, Cox N, et al. (2014) Spatially explicit trends in the global conservation status of vertebrates. PloS One 9(11):e113934. https://doi.org/10.1371/journal.pone.0113934
Roy BA, Zorrilla M, Endara L, Thomas DC, Vandegrift R, et al. (2018) New mining concessions could severely decrease biodiversity and ecosystem services in Ecuador. Trop Conserv Sci 11:194008291878042. https://doi.org/10.1177/1940082918780427
Salas-Molina F, Bistaffa F, Rodríguez-Aguilar JA (2023) A general approach for computing a consensus in group decision making that integrates multiple ethical principles. Socio-Econ Plan Sci 89:101694. https://doi.org/10.1016/j.seps.2023.101694
Sierra R, Calva O, Guevara A (2021) La Deforestación en el Ecuador, 1990-2018: Factores promotores y tendencias recientes. Ministerio de Ambiente y Agua del Ecuador; Ministerio de Agricultura del Ecuador, Quito, Ecuador
Taheripour F, Hertel TW, Ramankutty N (2019) Market-mediated responses confound policies to limit deforestation from oil palm expansion in Malaysia and Indonesia. Proc Nat Acad Sci United States Am 116(38):19193–19199. https://doi.org/10.1073/pnas.1903476116
Tanasescu M (2013) The rights of nature in Ecuador: the making of an idea. Int J Environ Stud 70(6):846–861. https://doi.org/10.1080/00207233.2013.845715
Tanner M, Ratzke L (2022) Deforestation, institutions, and property rights: evidence from land titling to indigenous peoples and local communities in Ecuador. CAF Banco de Desarrollo de Amérca Latina 22
Tapia-Armijos MF, Homeier J, Espinosa CI, Leuschner C, de La Cruz M (2015) Deforestation and forest fragmentation in South Ecuador since the 1970s - losing a hotspot of biodiversity. PloS One 10(9):e0133701. https://doi.org/10.1371/journal.pone.0133701
The World Bank (2023) GDP growth (annual %) - Ecuador. The World Bank. https://data.worldbank.org/indicator/NY.GDP.MKTP.KD.ZG?end=2020&locations=EC&start=1990 . Accessed 13 September 2023
United States Government (2016) Technical Support Document: Technical Update of the Social Cost of Carbon for Regulatory Impact Analysis - Under Executive Order 12866, pp 35
United States Government (2021) Technical Support Document: Social Cost of Carbon, Methane, and Nitrous Oxide: Interim Estimates under Executive Order 13990, 48 pp. https://www.whitehouse.gov/wp-content/uploads/2021/02/TechnicalSupportDocument_SocialCostofCarbonMethaneNitrousOxide.pdf . Accessed May 2023
Un-REDD programme (2016) Un-REDD programme Fact Sheet: About REDD+
Utreras R, Fierro LG, Viteri Mejía C (2017) Sostenibilidad Fiscal y Biodiversidaddel Ecuador. Polémika 12 (Año 5 - Semestre II), 93–115
Vallejo MC, Espinosa B, Venes F, López V, Anda S (2019) Esquivando estándares de desarrollo sustentable: Esudios de caso en proyectos hidroeléctricos del Ecuador, in: Ray, R., Gallagher, K., Sanborn, C. (Eds.), Development banks and sustainability in the Andean Amazon. Routledge, London, New York
Vanacker V, Molina A, Torres R, Calderon E, Cadilhac L (2018) Challenges for research on global change in mainland Ecuador. Neotropic Biodivers 4(1):114–118. https://doi.org/10.1080/23766808.2018.1491706
Vandegrift R, Thomas DC, Roy BA, Levy M (2018) The extent of recent mining concessions in Ecuador: a report detailing the overlap of exploratory mining concessions with protected forests and other regions of conservation and human rights interest in Ecuador. Rainforest Information Center
Vijay V, Pimm SL, Jenkins CN, Smith SJ (2016) The impacts of oil palm on recent deforestation and biodiversity loss. PloS One 11(7):e0159668. https://doi.org/10.1371/journal.pone.0159668
Weisse M, Potapov P (2021) Assessing trends in tree cover loss over 20 years of data. Global Forest Watch. https://www.globalforestwatch.org/blog/data-and-research/tree-cover-loss-satellite-data-trend-analysis/ . Accessed 12.2023
West TAP, Börner J, Sills EO, Kontoleon A (2020) Overstated carbon emission reductions from voluntary REDD+ projects in the Brazilian Amazon. Proc Nat Acad Sci United States Am 117(39):24188–24194. https://doi.org/10.1073/pnas.2004334117
West TAP, Wunder S, Sills EO, Börner J, Rifai SW, et al. (2023) Action needed to make carbon offsets from forest conservation work for climate change mitigation. Science (New York, N.Y.) 381(6660):873–877. https://doi.org/10.1126/science.ade3535
Download references
Open Access funding enabled and organized by Projekt DEAL. KP and JT: DFG-Project KN 586/19-1 “RESPECT”; MK: Horizon-EU-Project “WILDCARD”, Grant-Agreement-Nr. 101081177; LB: H2020-MSCA-ITN-2020 Project “Skill-For Action”, Grant-Agreement-Nr. 956355; JC: Fachagentur für nachwachsende Rohstoffe, Projekt “C-turn”, Förderkennzeichen 2218WK36X4.
Author information
Authors and affiliations.
Institute of Forest Management, Department of Life Science Systems, TUM School of Life Sciences Weihenstephan, Technical University of Munich, Hans-Carl-von-Carlowitz-Platz 2, Freising, 85354, Germany
Baltazar Calvas, Luz Maria Castro, Mengistie Kindu, Logan Bingham, Karla Pintado, Jonathan Torres Celi, Thomas Knoke & Jorge Cueva-Ortiz
Dirección General de Vinculación con la Sociedad, Universidad Técnica Particular de Loja, Loja, 1101608, Ecuador
Baltazar Calvas
Departamento de Economía, Universidad Técnica Particular de Loja, Loja, 1101608, Ecuador
Luz Maria Castro & Jonathan Torres Celi
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Jorge Cueva-Ortiz .
Ethics declarations
Conflict of interest.
The authors declare no competing interests.
Additional information
Communicated by David Lopez-Carr
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ .
Reprints and permissions
About this article
Calvas, B., Castro, L.M., Kindu, M. et al. Large differences between observed and expected Ecuadorian deforestation from 2001 to 2009: a counterfactual simulation approach. Reg Environ Change 24 , 94 (2024). https://doi.org/10.1007/s10113-024-02253-0
Download citation
Received : 25 January 2024
Accepted : 15 May 2024
Published : 01 June 2024
DOI : https://doi.org/10.1007/s10113-024-02253-0
How Does Deforestation Affect the Environment?

Forests, a vital component of life on Earth, cover approximately 31% of our planet’s land area . However, more than 75% of the Earth’s surface has been modified and degraded by human activities such as deforestation. Destroying forests alters weather patterns, destroys habitats, and negatively affects rural communities, leading to food insecurity and causing irreversible damage to entire ecosystems. So how does deforestation affect the environment and what threats does it pose to living species?
To answer the question of how deforestation affects the environment, it is important to look at why humans need forests in the first place. Deforestation is the purposeful cleaning of forest land for other uses. Among the main reasons for this damaging practice are agricultural expansion and cattle breeding as well as to obtain raw materials such as palm oil, a key ingredient in cosmetics and food products widely used around the world, and timber used for fuel, manufacturing, and infrastructure development. Studies show that 15,3 billion trees are chopped down every year and over the past 12,000 years, nearly 50% of the world’s trees have been purposefully cleared by humans. This practice threatens our environment, from altering the climate and various ecosystems to compromising the existence of millions of humans and animals.
You might also like: 10 Deforestation Facts You Should Know About
1. Effects on Climate Change
The scientific consensus on deforestation is that it intensifies climate change at a dramatic rate. The Global Forest Watch made it clear: protecting tropical rainforests is essential for achieving the climate goals of the Paris Agreement. Trees are known for their capacity to absorb carbon dioxide through photosynthesis. Healthy forests act as extremely valuable carbon sinks, with the Amazon rainforest being one of the world’s most important ones. However, deforestation is turning these sinks into huge net emitters , something that can have huge implications for slowing the pace of climate change and contributing to a steep rise in global temperatures. The current rate of rainforest-loss generated emissions is nearly 25% higher than those generated in the European Union and just slightly below US levels. Deforestation also increases the risk of uncontrollable wildfires because of humans burning vegetation. This, in turn, contributes to destroying forests, intensifying deforestation even more.
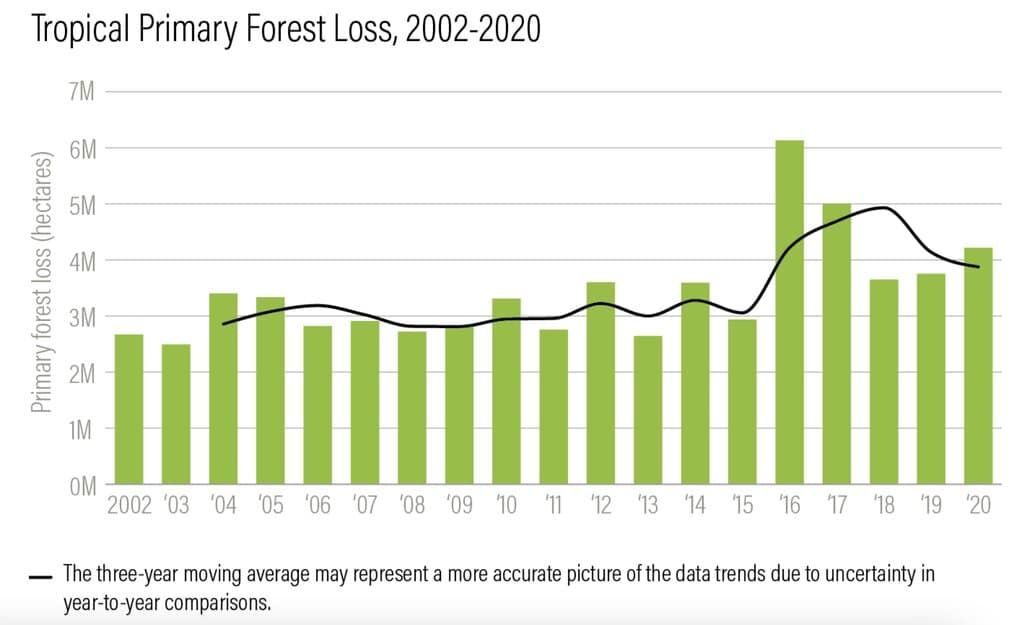
2. Effects on Soil Pollution and the Water Cycle
In addition to their role as carbon sinks, forests are a crucial component of the water cycle and have the all important function of preventing desertification. Cutting down trees can disrupt the cycle by decreasing precipitation and affecting river flow and water volume. In the case of the Amazon rainforest, research shows that at least 80% of its trees would be needed in order to keep the hydrological cycle going. With nearly 17% of the forest lost already, the Amazon is currently at its tipping point . Statistics show that deforestation in the tropics reduces precipitation over the Amazon by around 10% , or 138 millimeter, every year. In the South Asian Monsoon region, the reduction in rainfall is even higher, with around 18% less rain recorded in India in a single year.
Aside from their contribution to the water flow, trees help the land retain water and sustain forest life by supplying the soil with rich nutrients. Deforestation deprives the land of its cover, leaving the soil exposed to wind and rain. This makes soil vulnerable to being washed away, and prone to erosion. According to the World Wildlife Fund (WWF), as much as half of the world’s topsoil has been lost as a consequence of the nearly 4 million square miles of forest that have been lost since the beginning of the 20th century.
3. The Effects on Humans
In answering the question of how does deforestation affect the environment, you may discover that in fact, it also has a direct impact on the human population. With the loss of trees and entire forests, homelands are also being destroyed in the process. Indigenous communities who live in forests and depend on them to sustain their life bear the brunt of impacts from deforestation. As their houses are destroyed and resources compromised, these tribes are forced to migrate elsewhere and find other ways to sustain themselves. The Amazon rainforest is home to over one million Indigenous people , mostly of Indian descent, divided into more than 400 indigenous tribes. They live in settled villages by the rivers, and grow and hunt their food. These “uncontacted” tribes live by the rules of nature but are becoming increasingly vulnerable to deforestation, which has forced many of them to migrate. While some of them move into areas occupied by other tribes, straining the land’s resources, others are forced to relocate to urban settings and completely change their way of living.
4. The Effects on Animals and Plants
Along with Indigenous tribes, animals are some of the biggest victims of deforestation. Forests around the world are home to more than 80% of all terrestrial animal, plant, and insect species . However, the rapid destruction of forests is contributing to a decline in biodiversity never seen before. The main effect of deforestation on animals and plants is the loss of their habitat. Many factors related to cutting down trees contribute to driving species to extinction. Through land erosion, the soil is depleted of its nutrients, a huge source of nourishment for animals and plants. Furthermore, many animal species are heavily reliant on specific plants and their fruits for food sources. When these resources are lost, animals become weaker, more vulnerable to diseases and often succumb to starvation. Another important role of trees is to regulate the temperature of forests and maintain it constant. When deforestation occurs, temperature variates more drastically from day to night and this extreme change can often prove fatal for many animal species.
5. The Effects on Food Security
One last major effect of deforestation is its impact on food security through the loss of biodiversity. While food availability for Indigenous tribes and animals that live in forests is reduced in the process of deforestation, its effects on weather patterns and soil degradation also drastically decrease agricultural productivity. Populations located in the proximity of tropical forests are mostly impacted by the worsening trend. Indeed, millions of people living in these areas depend almost entirely on agriculture and are thus extremely vulnerable to the impact of deforestation on food security, struggling to grow enough food and prevent crops from damage. It has been shown that the deforestation of the Amazon contributes to a decline in pasture productivity of about 39% as well as a drop of soy yields of nearly 25% in over half of the Amazon region and of a staggering 60% in a third of the area.
You might also like: 12 Major Companies Responsible for Deforestation
Can We Halt Deforestation?
Knowing how deforestation affect the environment more than one way and its catastrophic effects on the planet, it is crucial that people around the world take action to mitigate its impact. This can be done on an individual level, for example by reducing meat consumption, going paperless and recycling products as much as possible, opting for natural products that do not contain ingredients such as palm oil and supporting organisations and sustainable companies that are committed to reducing this dangerous practice. On a governmental level, the consequences of deforestation can be mitigated by introducing policies that protect natural forests and regulate mining and logging operations as well as other operations that require the destruction of tree plantations.
Featured image: Global Water for Sustainability
This story is funded by readers like you
Our non-profit newsroom provides climate coverage free of charge and advertising. Your one-off or monthly donations play a crucial role in supporting our operations, expanding our reach, and maintaining our editorial independence.
About EO | Mission Statement | Impact & Reach | Write for us
About the Author

Martina Igini

10 of the World’s Most Endangered Animals in 2024

10 of the Most Endangered Species in India in 2024

The World’s Top 10 Biggest Rainforests
Hand-picked stories weekly or monthly. We promise, no spam!
- Name This field is for validation purposes and should be left unchanged.
Boost this article By donating us $100, $50 or subscribe to Boosting $10/month – we can get this article and others in front of tens of thousands of specially targeted readers. This targeted Boosting – helps us to reach wider audiences – aiming to convince the unconvinced, to inform the uninformed, to enlighten the dogmatic.
Case study - deforestation - the Amazon

Students also viewed

share this!
June 6, 2024
This article has been reviewed according to Science X's editorial process and policies . Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
reputable news agency
After historic floods, Brazil braces for severe drought

After torrential rains that sparked historic flooding in southern Brazil, the country expects a swing to severe drought in parts, the environment minister said Wednesday.
Brazil has been battered by a series of extreme weather events, most recently once-in-a-century flooding in the state of Rio Grande do Sul that left 172 people dead.
Environmental Minister Marina Silva said the flooding was due to a mash-up of natural phenomena such as El Niño and climate change, a double-whammy also seen elsewhere.
"We have the same thing happening in the Pantanal, the Amazon," she said, referring to climate extremes in one of the world's largest tropical wetlands, and its largest rainforest.
She said the northeastern Caatinga—a unique and biodiverse semi-arid biome—was "already experiencing moments of severe drought, and in the case of Rio Grande do Sul we are going to have severe drought ."
Silva, who was speaking at an event with President Luiz Inacio Lula da Silva, warned of more fires after record blazes in the first few months of the year.
A rapid study done by global scientists after the flooding in Rio Grande do Sul determined that climate change made the event twice as likely, with El Niño playing as big a role in the catastrophe.
El Niño, which alters rainfall patterns around the world, making parts more susceptible to torrential showers or drought, is currently weakening.
After a brief neutral period, La Niña—which can lead to drought conditions in parts of Latin America—is expected to return.
A renowned activist in her field, Silva returned to head the environment ministry and oversee Brazil's climate change policies when Lula returned to power in January 2023.
She had good news for Brazil's Cerrado region, a vast tropical savanna renowned for its rich biodiversity where deforestation soared 43 percent in 2023 while it halved in the Amazon.
Silva said that between January and May, deforestation had reduced 12.9 percent in the Cerrado but "it is too early to say that this is a lasting inflection in the curve."
Explore further
Feedback to editors

Researchers demonstrate new way to 'squeeze' infrared light
6 minutes ago

Elephants have names for each other like people do, new study shows
9 minutes ago
First detection of frost on the solar system's tallest volcanoes on Mars
Novel genetic clock discovers oldest known marine plant: Seagrass clone in the Baltic sea is more than 1,400 years old
18 minutes ago
Spliceosomes: New technique tracks proteins involved in RNA splicing
35 minutes ago
Compressed titanium and sulfur nanoribbons can transmit electricity without energy loss, scientists find
41 minutes ago

Protein study could help researchers develop new antibiotics
49 minutes ago

Siberia's 'mammoth graveyard' reveals 800-year human interactions with woolly beasts

Deriving mammalian DNA methylation predictors for maximum life span, gestation time and age at sexual maturity

New chip-scale erbium-based laser offers broad wavelength tunability
Relevant physicsforums posts, the secrets of prof. verschure's rosetta stones.
Jun 6, 2024
Is it possible to transform an electric thunderstorm into an EMP storm?
Jun 4, 2024
Jacchia Atmospheric Model
Jun 3, 2024
Iceland warming up again - quakes swarming
Mount ibu, indonesia erupts.
May 29, 2024
Adirondack Mountains and earthquakes
May 23, 2024
More from Earth Sciences
Related Stories

Deforestation in Brazil's Cerrado higher than in Amazon: Report
May 28, 2024

Deforestation hits record low in Brazilian Amazon in November
Dec 9, 2023

Deforestation down in Brazil's Amazon
Oct 7, 2023

Deforestation in Brazilian Amazon halved in 2023
Jan 5, 2024

Nearly 3,000 fires in Brazilian Amazon in February, new record
Feb 29, 2024

Deforestation in Brazilian Amazon down 22% in a year
Nov 10, 2023
Recommended for you

California wildfire pollution killed 52,000 in a decade: study
Jun 7, 2024

Wildfire smoke reached 99% of US lakes in 2019–2021: Study introduces 'lake-smoke day' metric

Scientists develop new method to estimate electrical parameters of regular pulse bursts in lightning

Faster alerts for California megaquakes: Early-warning system gets major upgrade
Coming in from the cold: Study reveals widespread negative experiences for women in polar research

New study finds Earth warming at record rate, but no evidence of climate change accelerating
Jun 5, 2024
Let us know if there is a problem with our content
Use this form if you have come across a typo, inaccuracy or would like to send an edit request for the content on this page. For general inquiries, please use our contact form . For general feedback, use the public comments section below (please adhere to guidelines ).
Please select the most appropriate category to facilitate processing of your request
Thank you for taking time to provide your feedback to the editors.
Your feedback is important to us. However, we do not guarantee individual replies due to the high volume of messages.
E-mail the story
Your email address is used only to let the recipient know who sent the email. Neither your address nor the recipient's address will be used for any other purpose. The information you enter will appear in your e-mail message and is not retained by Phys.org in any form.
Newsletter sign up
Get weekly and/or daily updates delivered to your inbox. You can unsubscribe at any time and we'll never share your details to third parties.
More information Privacy policy
Donate and enjoy an ad-free experience
We keep our content available to everyone. Consider supporting Science X's mission by getting a premium account.
E-mail newsletter
Living World - Amazon Case Study
The Amazon is the largest tropical rainforest on Earth. It sits within the Amazon River basin, covers some 40% of the South American continent and as you can see on the map below includes parts of eight South American countries: Brazil, Bolivia, Peru, Ecuador, Colombia, Venezuela, Guyana, and Suriname. The actual word “Amazon” comes from river.

Amazing Amazon facts; • It is home to 1000 species of bird and 60,000 species of plants • 10 million species of insects live in the Amazon • It is home to 20 million people, who use the wood, cut down trees for farms and for cattle. • It covers 2.1 million square miles of land • The Amazon is home to almost 20% of species on Earth • The UK and Ireland would fit into the Amazon 17 times!
The Amazon caught the public’s attention in the 1980s when a series of shocking news reports said that an area of rainforest the size of Belgium was being cut down and subsequently burnt every year. This deforestation has continued to the present day according to the Sao Paulo Space Research Centre. In 2005 they had lost 17% of Amazon rainforest or 650000 square kilometres. Their satellite data is also showing increased deforestation in parts of the Amazon. The process of deforestation The Amazon helps a Newly Emerging Economy(NEE), Brazil, to make money. They build roads into the forest, logging firms then go in and take out valuable hard woods such as mahogany and cedar, worth thousands of pounds in richer economies like Europe. Then farmers, often cattle ranchers from big companies, burn the rest to make way for cattle pasture. 75% of cleared areas are used in this way. This is clearly shown on the map on figure 22 in red. Many of the deforested areas follow roads and branch off from there. Deforestation is also worse in the South and South East of the Amazon basin, closer to major centres of population in Brazil.

© WWF Source Used with permission.
The causes of deforestation 1. Subsistence and commercial farming – subsistence farming is where poor farmers occupy plots of the forest to grow food to feed themselves and their families. They clear forest and then burn it, hence the name slash and burn. They grow crops until the soil is exhausted and then move on. This contributes to deforestation but not as much as commercial farming (Farming to sell produce for a profit to retailers or food processing companies). The Brazilian region of Mato Grosso was affected by deforestation in the 1980s and 1990s. 43% of rainforest losses were in this region, and area almost ½ the size of France. It has been replaced by fields for grain and cattle. This has allowed Brazil to overtake Australia as the largest exporter of beef in the world. The land is also flat and easy to farm. It also has high temperatures and lots of rainfall.
2. Logging – This involves cutting down trees for sale as timber or pulp. The timber is used to build homes, furniture, etc. and the pulp is used to make paper and paper products. Logging can be either selective or clear cutting. Selective logging is selective because loggers choose only wood that is highly valued, such as mahogany. Clear-cutting is not selective. Loggers are interested in all types of wood and therefore cut all of the trees down, thus clearing the forest, hence the name- clear-cutting.
3. Road building – trees are also clear for roads. Roads are an essential way for the Brazilian government to allow development of the Amazon rainforest. However, unless they are paved many of the roads are unusable during the wettest periods of the year. The Trans Amazonian Highway has already opened up large parts of the forest and now a new road is going to be paved, the BR163 is a road that runs 1700km from Cuiaba to Santarem. The government planned to tarmac it making it a superhighway. This would make the untouched forest along the route more accessible and under threat from development.
4. Mineral extraction – forests are also cleared to make way for huge mines. The Brazilian part of the Amazon has mines that extract iron, manganese, nickel, tin, bauxite, beryllium, copper, lead, tungsten, zinc and gold!

The Belo Monte dam site under construction, copyright Used with the kind permission of Phil Clarke-Hill - His website is amazing, click here to see it.
5. Energy development – This has focussed mainly on using Hydro Electric Power, and there are 150 new dams planned for the Amazon alone. The dams create electricity as water is passed through huge pipes within them, where it turns a turbine which helps to generate the electricity. The power in the Amazon is often used for mining. Dams displace many people and the reservoirs they create flood large area of land, which would previously have been forest. They also alter the hydrological cycle and trap huge quantities of sediment behind them. The huge Belo Monte dam started operating in April 2016 and will generate over 11,000 Mw of power. A new scheme the 8,000-megawatt São Luiz do Tapajós dam has been held up because of the concerns over the impacts on the local Munduruku people.

6. Settlement & population growth – populations are growing within the Amazon forest and along with them settlements. Many people are migrating to the forest looking for work associated with the natural wealth of this environment. Settlements like Parauapebas, an iron ore mining town, have grown rapidly, destroying forest and replacing it with a swath of shanty towns. The population has grown from 154,000 in 2010 to 220,000 in 2012. The Brazilian Amazon’s population grew by a massive 23% between 2000 and 2010, 11% above the national average.
Impacts of deforestation – economic development, soil erosion, contribution to climate change. • Every time forest is cleared species are lost – so we lose BIODIVERSITY • Climate Change - Burning the forest releases greenhouse gasses like CO2. This contributes to the warming of our planet via climate change and global warming. In addition, the loss of trees prevents CO2 being absorbed, making the problem worse. The Amazon also helps to drive the global atmospheric system. There is a lot of rainfall there and changes to the Amazon could disrupt the global system. • Economic development – Brazil has used the forests as a way to develop their country. The forest has many natural riches that can be exploited. In addition, Brazil has huge foreign debt and lots of poor people to feed, so they want to develop the forest. May Brazilians see deforestation as a way to help develop their country and improve people’s standard of living. • Soil erosion - the soils of the Amazon forest are not fertile and are quickly exhausted once the forest is cleared. The farmers now artificially fertilise the soil when in the past the nutrient cycle would have done this naturally. In addition, the lack of forest cover means that soils are exposed to the rainfall. This washes huge amounts of soil into rivers in the process of soil erosion.
NEXT TOPIC - Living World - Sustainable Forest Management
©2015 Cool Geography
- Copyright Policy
- Privacy & Cookies
- Testimonials
- Feedback & support


IMAGES
VIDEO
COMMENTS
A new study, co-authored by a team of researchers including UConn Ecology and Evolutionary Biology researcher Cory Merow provides the first quantitative assessment of how environmental policies on deforestation, along with forest fires and drought, have impacted the diversity of plants and animals in the Amazon.
The Amazon in context. Tropical rainforests are often considered to be the "cradles of biodiversity.". Though they cover only about 6% of the Earth's land surface, they are home to over 50% of global biodiversity. Rainforests also take in massive amounts of carbon dioxide and release oxygen through photosynthesis, which has also given ...
Date: November 08, 2022. The numbers are devastating: 17% of Amazon forests have been wholly lost, and an additional 17% are degraded. And data from the first half of 2022 show the loss continuing to grow. The Amazon is in crisis as forests are threatened by deforestation, fires, and degradation; surface water has been lost; and rivers are ...
Based on estimates of 1% annual tropical forest loss, the Amazon may be losing as many as 11 to 16 species per day (Wilson 1989), and the resulting ecosystems are often highly degraded (Buschbacher 1986). Te deforestation of Amazonia presents a challenging study of the interactions among people, their values, and the environment.
The sector is responsible for deforestation in the Amazon, cases of work similar to slavery, and is also relevant in the country's economy, both for meat, as we are the largest exporter of meat, and also for the export of biodiesel, which is made with animal fat, and leather, from clothing to automobiles. ... Case Studies Member Profiles ...
Approximately 20% of the Brazilian Amazon has now been deforested, and the Amazon is currently experiencing the highest rates of deforestation in a decade, leading to large-scale land-use changes. Roads have consistently been implicated as drivers of ongoing Amazon deforestation and may act as corridors to facilitate species invasions.
Semantic Scholar extracted view of "Spatiotemporal Assessment of Deforestation and Forest Degradation Indicates Spillover Effects From Mining Activities and Related Biodiversity Offsets in Madagascar" by Sandra Eckert et al. ... Approximately 2.5 × 106 square kilometers of the Amazon forest are currently degraded by ... Case Study in Inner ...
Studies rarely take both climate change and deforestation into account. But the new study's results bolster the findings of other scientists who have modeled results showing that when the Amazon ...
CASE STUDY Category: ICTs and Climate Change Monitoring private lands) in the Amazon from 50 to 80% in 1996; the creation of the plan to protect and control defores tation in the Amazon (PPCDAM) in 2004; the creation of decree 6321/2007 limiting bank credit
Approximately 20% of the Brazilian Amazon has now been deforested, and the Amazon is currently experiencing the highest rates of deforestation in a decade, leading to large-scale land-use changes. Roads have consistently been implicated as drivers of ongoing Amazon deforestation and may act as corridors to facilitate species invasions.
The stratigraphy of core KET 8003 (38°49.2' N, 14°29.5' E, 1,900 m water depth, 1,030 cm length), is based on the oxygen isotope variation in the foraminifer Globigerina bulloi'des, measured at ...
This study, therefore, both generalizes spatially and formalizes existing knowledge on the causes of deforestation that was gained through local scale case studies. The analytical approach adopted in this study scales up findings from the local to the regional scale without altering conclusions, thus allowing researchers to get round the so ...
deforestation in the 1990s ( 10). Similar or greater amounts may be held in soil carbon, but these are less vulnerable to loss after deforestation (20). In addition, forest plot studies suggest that intact forests are a carbon sink (~0.6 Pg C year -1) (21), particularly in more fertile western Ama-zonia. The existence of this sink is debated ...
The Deforestation of the Amazon: A Case Study in Understanding Ecosystems and Their Value Part II: Data Analysis and Perspectives Worksheet By: Phil Camill, Department of Biology Carleton College, Northfield, MN The Problem At the frontier of primary Amazonian rainforest, a five-hectare plot is under consideration for ...
Biodiversity contributes to the ecological and climatic stability of the Amazon Basin 1, 2, but is increasingly threatened by deforestation and fire 3, 4. Here we quantify these impacts over the ...
They will complement this archival research with field interviews and intensive case-studies involving at least five landowners and many local key informants. ... globally associated with agrarian reform, has waxed and waned in the Brazilian Amazon over the past three decades, a period that witnessed high rates of deforestation and hundreds of ...
Case Study: Losing the Rainforest, One Tree at a Time. Studying Change over Time. ... satellite images are used to study change in the Amazon rainforest. There are three great reasons for using SAR images to look at the rainforest. ... deforestation accounts for approximately 20% of global emissions. This is more than the amount released by 13 ...
disseminante the findings of these case studies, and stimulate further studies in the area of climate impacts. A seminar entitled "Hydrology and Water Management of the Amazon Basin" was held in Manaus, Brazil on 5-9 August 1990 and sponsored by the Institute for Superior Studies of the Amazon and Brazilian Association of Hydraulic Resources.
Abstract. In this case study, students examine tropical deforestation in the Amazon from the perspective of three dominant stakeholders in the region: a peasant farmer, logger, and environmentalist. As part of the exercise, students perform a cost-benefit analysis of clearing a plot of tropical forest in the Amazon from the perspective of one ...
Corpus ID: 146632308; The Deforestation of the Amazon : A Case Study in Understanding Ecosystems and Their Value @article{Camill1999TheDO, title={The Deforestation of the Amazon : A Case Study in Understanding Ecosystems and Their Value}, author={Phillip Camill and Ruth Ann Althaus and Robert Boyd Skipper and Heidi Malm and Kendra Spence Cheruvelil and Patricia A. Soranno}, journal={Science ...
The impact of agricultural colonization and deforestation on orchid bees (Apidae: Euglossini) in the Brazilian Amazon. Biological Conservation , 293 , 110560. doi: 10.1016/j.biocon.2024.110560
Deforestation in Brazil's Amazon rainforest has hit its highest level in over 15 years, official data shows. A report by Brazil's space research agency (Inpe) found that deforestation increased by ...
Ensuring the integrity of the world's forests is indispensable for mitigating climate change, combatting biodiversity loss, and protecting the livelihoods of rural communities. While many strategies have been developed to address deforestation across different geographic scales, measuring their impact against a fluctuating background of market-driven forest loss is notoriously challenging ...
The Deforestation of the Amazon: A Case Study in Understanding Ecosystems and Their Value. Phil Camill. 1999. Bibliographic information. Title: The Deforestation of the Amazon: A Case Study in Understanding Ecosystems and Their Value: Author: Phil Camill: Published: 1999 : Export Citation:
3. The Effects on Humans. In answering the question of how does deforestation affect the environment, you may discover that in fact, it also has a direct impact on the human population. With the loss of trees and entire forests, homelands are also being destroyed in the process. Indigenous communities who live in forests and depend on them to ...
Amazon Deforestation: A Regional Conservation Case Study. GIS analysis of select strictly protected areas supported by the Amazon Region Protected Areas Program (ARPA)
There are lots of causes of deforestation of the Amazon - for example between 2000 and 2005 (fill in blanks)65-70% - a20-25% -b5-10%- c1-2% - d. A - caused by commercial (cattle) ranching.B - small scale subsistence farming.C - logging, illegal logging.D - other activities like mineral extraction, road building, energy development and building ...
Silva said that between January and May, deforestation had reduced 12.9 percent in the Cerrado but "it is too early to say that this is a lasting inflection in the curve." After torrential rains ...
The actual word "Amazon" comes from river. Amazing Amazon facts; • It is home to 1000 species of bird and 60,000 species of plants • 10 million species of insects live in the Amazon • It is home to 20 million people, who use the wood, cut down trees for farms and for cattle. • It covers 2.1 million square miles of land • The ...