
Article & Observations About Diverse Teams Working Better Together

Software Testing Client Project Case Study
Apr 21 • Case Studies
We are often asked what software testing is . The video below shares a solid definition of the term.
But we thought a software testing project case study might be helpful to better understand what software testers do on a typical day. This includes testing software, writing requirement documents for our clients, and creating user guides to ensure compliance for our clients to use for quality assurance and auditing purposes.
Iterators LLC was hired to complete accessibility testing for a few projects for the Library of Congress (LOC). Accessibility testing is required on all government websites, with Section 508 and WCAG 2.2 requirements used. To become a Trusted Tester an employee must complete the DHS Trusted Tester online training course requirements and pass the DHS Section 508 Trusted Tester Certification Exam of Homeland Security in Accessibility, and we are in a unique position to help on this project. We cross-train all our employees so that we can work on several projects at one time or several different aspects of a project to complete the work and reduce the cost to our clients.
Our first project assigned by LOC was testing their new braille feature on BARD Mobile for Android. We were tasked with testing the braille feature with several refreshable braille displays.
During our testing, we used the Orbit Reader 20 , and two different braille displays from Freedom Focus 14 and Freedom Focus 40 . There are plans to use other refreshable displays such as Humanware, but this testing has not occurred yet. We needed to test refreshable braille displays and their use in tandem with Google BrailleBack and Google TalkBack .
This work was to ensure that all hardware worked as expected with the apps we were testing. For this testing, we had to complete functional testing, smoke testing, exploratory testing and had a user panel to ensure we caught all issues that a visually impaired individual might experience while using the app.
Initially, our client was unsure if we would find any bugs and hesitant to have us enter bugs into Bugzilla as they stated the software was “complicated”. Bugzilla is a web-based general-purpose bug tracking system and not unlike other bug tracking systems we use every day such as Jira, TestRails, PractiTest, and Click-Up.
Testing was completed over several agile sprints with many significant software testing bugs found. Our testing had us test against the National Library Service requirements document. Next, we had to create an up-to-date user manual. While the manual had been updated several times, the testing had not been.
For example, when downloading a book or magazine from the Now Reading section of the mobile app, the download would end up at the bottom of the page. For years, the user guide had listed the download being at the top of the page once the document was downloaded.
Our testing team, on several occasions, said this was an error in the documentation and that the download ends up at the bottom of the page. This was corrected in the user document and sent to the development team to fix per the requirement document.
Over the next several months, we reported 30 high-priority bugs with about half fixed at this point. We have encouraged our client to test in an agile fashion because once the development team is finished, it’s harder to get these bugs fixed.
Our bugs were reported and based on the requirement document used to create the software. Lastly, the user guide had to be rewritten to reflect the app's behavior and general updates.
Once the app was tested and created, the user guide was sent to Communication Services to ensure the style used per other requirement documentation. This document had to be approved before being disseminated to the public. For example, how does the library determine what the Most Popular Books are, and over what period.
Once the document was returned from COS, this PDF document had to be remediated . The process of remediation is to make a PDF, create the heading for the document, alt text added to meaningful images, and decorative images either ignored or taken out of the digital document altogether.
Once the remediation process is complete and validated, the document becomes ADA-compliant. We then provide an accessible PDF that can be read with the use of a screen reader and create the HTML output so that the document can be added to the Library of Congress website.
You can find the current user guide we completed here: https://www.loc.gov/nls/braille-audio-reading-materials/bard-access/bard-mobile-android/#creatingfolders3.3
Case studies can be a great learning tool in software testing and project management. By looking at project case study examples, you can see how the project was planned and executed, as well as how certain tasks were managed. This can give a better understanding of what software testing involves on a daily basis. With the right software testing case studies example, software testers can hone their skills, improve project performance, and ultimately deliver better software testing results.
Related Resources:
- Crafting an Effective Test Plan: A Step-by-Step Guide
- Top Test Management Tools
- Mobile Application Functional and Performance Testing
About the Author
Jill Willcox has worked on accessibility issues for most of her professional career. Iterators is an inclusive women-owned small business (WOSB) certified by the Small Business Administration and WBENC. We provide software testing services for websites, mobile apps, enterprise software, and PDF remediation services, rendering PDFs ADA compliant.
Jill Willcox
Clutch names iterators llc as a top certified women-owned business for 2022.
Iterators LLC named Top Certified Women0owned Business Again 2022
Test Strategy vs Test Plan: What’s the Difference?
What is the difference between a test strategy and a test plan? Read this article to b...
May 05 • Reference
- Test Automation
- Performance Engineering and Testing
- System Integration Testing Service
- Security Testing
- Functional Testing Services
- Data Validation
- Mobile Testing Service
- Audit & Advisory Services
- Emerging Technologies
- On Demand Testing™
- Accessibility Testing
- Accessibility WCAG Training
- Translation and Localization Services
- Digital Media
- Banking and Financial
- Construction and Industrial
- Media and Entertainment
- Retail Software Testing
- Telecommunications
- Energy and Utilities
- Hospitality & Tourism
- Our Clients
Case Studies
- White Papers
- Leadership Team
- Working at QA Consultants
- Current Opportunities
Client Success Stories – See who we’ve helped.
QA Consultants helped Toronto Hydro
CASE STUDIES
Privacy overview.
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |

QA Case Study
- Industry Financial
- Solution E-mail platform build automation
- Company The customer needed an outsourced QA team to start manual testing of their products. As an established business Client required full-time QA engineers in-house so they needed a company that had experts in place that they could use.
- Tools & platforms Usually startups are less concerned
Software Quality Assurance Case Study
QA case study : Usually startups are less concerned about the quality and in many cases developers are doing QA part of work, we understand that it saves cost and gives chance to release product in a short period of time but on another hand clients will face poor quality this is exactly the situation we saw happened with our customer in Vancouver – financial technology company was looking for a way how to improve their quality and decided to find an outsourcing partner in order to set up QA team from scratch. After RFP process – GeeksForLess been chosen as a strategic partner in order to start manual testing and then move towards automation testing.
Solution we made
- SEPARATED TEST CASES PER APPLICATIONS AND PROJECTS
- COVERED 99% OF ALL REQUIREMENTS AND ALREADY EXISTING FUNCTIONALITY WITH TEST CASES
- ADDED BASIC TEST PLANS
- AUTOMATED ALL TEST CASES WHICH ARE STABLE AND COULD BE COVERED BY JS TOOL
- ESTABLISHED PROCESS – NO MORE RELEASES TO PRODUCTION WITHOUT QA CONFIRMATION
- SETUP DAILY CALLS WITH UPDATES ON THE PROGRESS
- MOVED TOWARDS CONTINUOUS INTEGRATION BY USING SPECIAL TOOLS, AUTOTESTS WERE RUNNING AUTOMATICALLY AFTER PRODUCTION RELEASES
- QA ENVIRONMENT HAS BEEN SET UP INTEGRATED QA TEAM INTO ALL PROJECT ASPECTS SO THEY BECAME A FULL PART OF CLIENT ECO-SYSTEM CUSTOMERS
or check our contact info

- Onsite training
3,000,000+ delegates
15,000+ clients
1,000+ locations
- KnowledgePass
- Log a ticket
01344203999 Available 24/7

Software Testing Case Studies: A Comprehensive Analysis
Software Testing Case Studies offer real-world insights into how companies identify and solve software issues, ensuring quality and reliability. This blog delves into various case studies, showcasing different testing methodologies, challenges faced, and solutions implemented. Read more to explore them in detail!

Exclusive 40% OFF
Training Outcomes Within Your Budget!
We ensure quality, budget-alignment, and timely delivery by our expert instructors.
Share this Resource
- ISTQB Advanced Level Test Manager
- ISTQB Advanced Level Test Analyst
- ISTQB Agile Foundation Extension
- ISTQB Advanced Test Automation Engineer
- ISTQB Advanced Level Technical Test Analyst

Have you ever wondered how leading companies ensure their software is reliable and bug-free? Or what critical process they perform to check if a software application or system meets specified requirements. In this blog, we will discuss some real-world Software Testing Case Studies to uncover the strategies that drive successful testing outcomes.
Such examples will help you identify bugs, errors, and issues to ensure the software is reliable, secure and performs as expected. By thoroughly examining the Software Testing Case Studies, you can receive valuable knowledge on best practices, common challenges, and innovative solutions in Software Testing. Ready to enhance your understanding and elevate your testing game? Let’s dive in.
Table of Contents
1) Software Testing Case Studies
a) Example 1: Banking Software Enhancement at Barclays
b) Example 2: E-commerce Platform Optimisation at ASOS
c) Example 3: Government Digital Services Improvement
2) Conclusion
Software Testing Case Studies
Exploring real-world examples of Software Testing can give detailed insights into best practices and common challenges. These case studies highlight how different organisations have approached Software Testing to ensure quality and reliability. By examining these examples, you can gain a deeper understanding of effective testing strategies and methodologies.
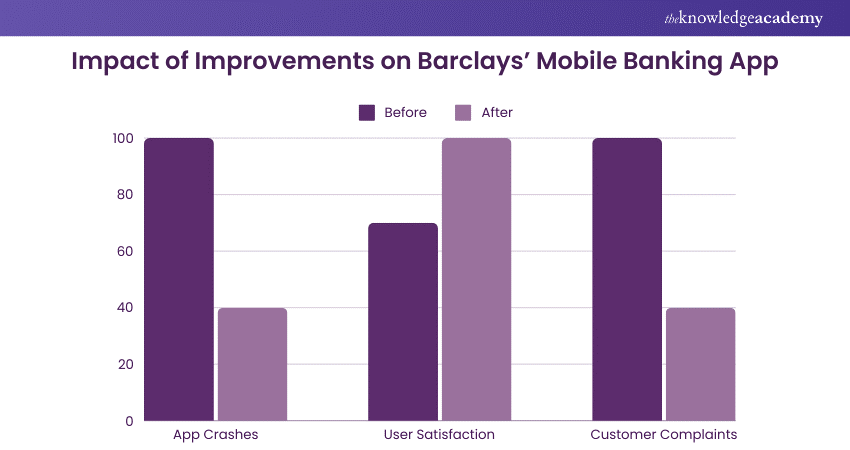
Example 1: Banking Software Enhancement at Barclays
Company Overview:
Barclays, one of the UK's leading multinational banks, offers a huge range of financial services, that include personal banking, corporate banking, wealth management, and investment banking.
The Challenge:
Barclays' mobile banking app was experiencing a high rate of crashes and performance issues during peak usage times. Customers were frustrated by frequent downtimes and transaction errors, impacting the bank's reputation for reliability.
Objectives:
1) Improve the stability and performance of the mobile banking app
2) Reduce the number of customer complaints on app crashes
3) Ensure seamless and secure user experience during high-traffic periods
Approach:
1) Assessment and Planning:
a) Conducted a comprehensive review of the existing testing processes
b) Developed a detailed testing strategy focusing on performance and load testing.
2) Test Automation:
a) Introduced automated testing for critical functions of the mobile app
b) Created automated scripts to test various user scenarios and transactions
3) Performance and Load Testing:
a) Conducted extensive load testing to simulate peak usage conditions
b) Identified and addressed performance bottlenecks
4) Continuous Testing:
a) Implemented continuous integration and continuous testing (CI/CT) to ensure new updates were thoroughly tested before release.
Results:
a) Reduced app crashes by 60%
b) Improved app performance, leading to a 30% increase in user satisfaction
c) Decreased customer complaints regarding app issues, enhancing the bank's reputation.
Boost your Agile Testing skills with our ISTQB Agile Foundation Extension Training - secure your spot today!
Example 2: E-commerce Platform Optimisation at ASOS
AsSeenOnScreen (ASOS), a leading UK-based online fashion retailer, caters to customers worldwide with a vast range of clothing, accessories, and beauty products.
ASOS faced challenges with its website's performance during major sales events. High traffic volumes led to slow page loads and transaction failures, resulting in lost sales and customer dissatisfaction.
a) Ensure the website can handle high traffic volumes during sales events
b) Improve overall website performance and user experience
c) Minimise transaction failures and abandoned carts
a) Analysed the existing infrastructure and identified key performance issues
b) Developed a testing plan focusing on load and stress testing
2) Load and Stress Testing:
a) Simulated high-traffic conditions to identify potential points of failure
b) Implemented solutions to enhance the website's scalability and resilience
3) Performance Optimisation:
a) Optimised backend processes and database queries to improve response times
b) Streamlined the front-end code for faster page loads
4) Continuous Monitoring:
a) Established real-time monitoring to track website performance during peak times.
b) Quickly addressed any issues that arose during high-traffic periods
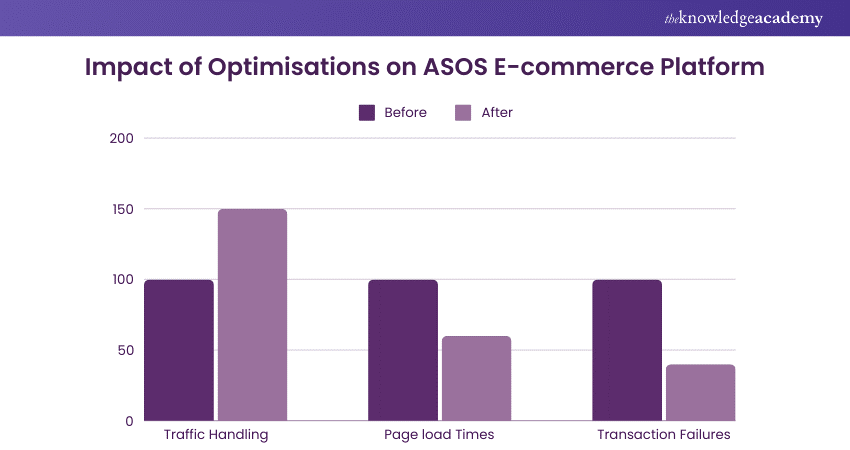
a) The website handled a 50% increase in traffic without performance degradation
b) Page load times improved by 40%, enhancing the user experience
c) Significant reduction in transaction failures, leading to higher sales conversion rates.
Become a Certified Tester with our ISTQB Software Testing Foundation Course - register now and boost your skills!
Example 3: Government Digital Services Improvement
Organisation Overview:
The UK Government Digital Service (GDS) is responsible for digital transformation across government departments, ensuring public services are simple, efficient, and user-friendly.
Several government websites and digital services experienced frequent downtime and security vulnerabilities, impacting public access to essential services.
a) Enhance the reliability and security of government digital services
b) Improve user accessibility and experience
c) Guarantee compliance with Data Protection and Cyber Security standards
a) Conducted a thorough assessment of existing digital services
b) Developed a comprehensive testing strategy focusing on security and accessibility
2) Security Testing:
a) Implemented rigorous security testing to identify and mitigate vulnerabilities
b) Conducted regular penetration testing and security audits
3) Accessibility Testing:
a) Ensured all digital services met UK accessibility standards
b) Conducted user testing with diverse groups to identify and address accessibility issues
4) Continuous Improvement:
a) Established a continuous improvement framework to regularly update and enhance digital services.
b) Implemented real-time monitoring to quickly identify and resolve issues.
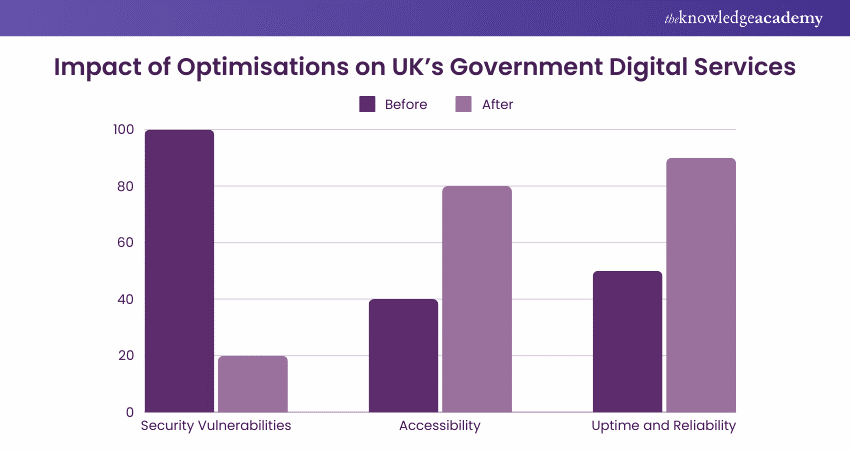
a) Enhanced security posture, reducing vulnerabilities by 80%
b) Improved accessibility, making services easier for all citizens to use
c) Increased uptime and reliability, leading to higher user satisfaction and trust in government services.
Master Test Management skills with our ISTQB Advanced Level Test Manager Course - book your spot now!
Conclusion
Software Testing Case Studies provide a treasure trove of insights into effective testing practices and innovative solutions. By learning from these real-world examples, you can significantly enhance your testing strategies. Alongside this, it will help you ensure superior software quality.
Advance your testing expertise with our ISTQB Advanced Test Analyst Course - sign up now!
Frequently Asked Questions
A good software test case is clear, concise, and comprehensive. It specifies the input, expected output, and steps to execute. It covers positive and negative scenarios, ensuring thorough validation of the software's functionality.
The Knowledge Academy takes global learning to new heights, offering over 30,000 online courses across 490+ locations in 220 countries. This expansive reach ensures accessibility and convenience for learners worldwide.
Alongside our diverse Online Course Catalogue, encompassing 17 major categories, we go the extra mile by providing a plethora of free educational Online Resources like News updates, Blogs , videos, webinars, and interview questions. Tailoring learning experiences further, professionals can maximise value with customisable Course Bundles of TKA .
The Knowledge Academy’s Knowledge Pass , a prepaid voucher, adds another layer of flexibility, allowing course bookings over a 12-month period. Join us on a journey where education knows no bounds.
The Knowledge Academy offers various Software Testing Courses , including the ISTQB Agile Foundation Extension Course, Certified Software Testing Professional (CSTP) Course, and Unit Testing Course. These courses cater to different skill levels, providing comprehensive insights into Software Unit Testing .
Our Business Analysis Blogs cover a range of topics related to Software Testing, offering valuable resources, best practices, and industry insights. Whether you are a beginner or looking to advance your Software Testing skills, The Knowledge Academy's diverse courses and informative blogs have got you covered.
Upcoming Business Analysis Resources Batches & Dates
Mon 29th Jul 2024
Mon 12th Aug 2024
Tue 27th Aug 2024
Mon 9th Sep 2024
Mon 23rd Sep 2024
Mon 7th Oct 2024
Mon 21st Oct 2024
Mon 4th Nov 2024
Mon 18th Nov 2024
Mon 2nd Dec 2024
Mon 16th Dec 2024
Mon 6th Jan 2025
Mon 20th Jan 2025
Mon 3rd Feb 2025
Mon 17th Feb 2025
Mon 3rd Mar 2025
Mon 17th Mar 2025
Mon 31st Mar 2025
Mon 28th Apr 2025
Mon 19th May 2025
Mon 2nd Jun 2025
Mon 16th Jun 2025
Mon 30th Jun 2025
Mon 14th Jul 2025
Mon 28th Jul 2025
Mon 11th Aug 2025
Mon 8th Sep 2025
Mon 22nd Sep 2025
Mon 6th Oct 2025
Mon 20th Oct 2025
Mon 3rd Nov 2025
Mon 17th Nov 2025
Mon 1st Dec 2025
Mon 15th Dec 2025
Get A Quote
WHO WILL BE FUNDING THE COURSE?
My employer
By submitting your details you agree to be contacted in order to respond to your enquiry
- Business Analysis
- Lean Six Sigma Certification
Share this course
Our biggest spring sale.
* WHO WILL BE FUNDING THE COURSE?
We cannot process your enquiry without contacting you, please tick to confirm your consent to us for contacting you about your enquiry.
By submitting your details you agree to be contacted in order to respond to your enquiry.
We may not have the course you’re looking for. If you enquire or give us a call on 01344203999 and speak to our training experts, we may still be able to help with your training requirements.
Or select from our popular topics
- ITIL® Certification
- Scrum Certification
- Change Management Certification
- Business Analysis Courses
- Microsoft Azure Certification
- Microsoft Excel Courses
- Microsoft Project
- Explore more courses
Press esc to close
Fill out your contact details below and our training experts will be in touch.
Fill out your contact details below
Thank you for your enquiry!
One of our training experts will be in touch shortly to go over your training requirements.
Back to Course Information
Fill out your contact details below so we can get in touch with you regarding your training requirements.
Preferred Contact Method
No preference
Back to course information
Fill out your training details below
Fill out your training details below so we have a better idea of what your training requirements are.
HOW MANY DELEGATES NEED TRAINING?
HOW DO YOU WANT THE COURSE DELIVERED?
Online Instructor-led
Online Self-paced
WHEN WOULD YOU LIKE TO TAKE THIS COURSE?
Next 2 - 4 months
WHAT IS YOUR REASON FOR ENQUIRING?
Looking for some information
Looking for a discount
I want to book but have questions
One of our training experts will be in touch shortly to go overy your training requirements.
Your privacy & cookies!
Like many websites we use cookies. We care about your data and experience, so to give you the best possible experience using our site, we store a very limited amount of your data. Continuing to use this site or clicking “Accept & close” means that you agree to our use of cookies. Learn more about our privacy policy and cookie policy cookie policy .
We use cookies that are essential for our site to work. Please visit our cookie policy for more information. To accept all cookies click 'Accept & close'.
- +1 (469) 891-7117
- [email protected]
Case Studies
- Let's Connect
- Book a Call
Home > Case Studies
- API Testing
- Automation Testing
- Cloud Testing
- Compatibility Testing
- Content QA Testing
- Embedded Testing
- Functional Testing
- Integration Testing
- Load Testing
- Mobile Application Testing
- Performance Testing
- Regression Testing
- Sanity testing
- Security Testing
- Software Testing
- System Testing
- Usability Testing
- Vulnerabilities Testing
- Electronics
- Health and Wellness
- Logistics and Supply Chain
- Manufacturing
- Retail and E-commerce
SAP CRM Performance Testing Solution for India’s Largest Natural Gas Distribution Company
Deployed advanced testing strategies to elevate product quality for a leading it management software provider, reduced software errors 75% faster with end-to-end testing for a leading north american real estate technology provider, revolutionizing restaurant operations: vita mojo's digital transformation with impactqa, supported india's second-largest two-wheeler company in eliminating compatibility and performance bottlenecks, successfully automated workflows for kpmg to ensure process optimization, efficiently managed end-to-end regression workflows for clubspark to achieve 100% automation coverage , improve user experience by achieving 60% better app performance for a leading global beverages retail/online store, expert testing ability to improve website performance & load capability by 70% for new york road runners (nyrr), get started..
Experience a Free 30 Min Consultation with an Expert.
Schedule a Free Consultation
Subscribe to our newsletter
Get the latest industry news, case studies, blogs and updates directly to your inbox
Invalid Captcha!
Case studies
Cybersecurity Assessment for a Leading Bank in a GCC Country
Exploratory and Security Testing For a Virtual Space Solution
Accessibility and Functional Testing For Interactive Video Platform
End-to-End Testing for the AI-Powered Meeting Platform
Manual and Automated Testing for the Mental Health Support Application
Testing of Cross-Platform SDK for Healthcare Developers
We use cookies to ensure that we give you the best experience on our website. If you continue to use this site we will assume that you are happy with it.
Privacy Overview
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
Case Studies
Discover our software testing case studies that show how we've helped top brands achieve their business and software development goals..
- Mobile Testing
- Performance Testing
- Software Testing
- Test Automation
- Tool Development
- AI Development

We innovatively crafted an AI copilot for Bantotal, named Bantotal Copilot.

We helped Shutterfly to maintain its site's competitive advantage of delivering a superior customer experience.

We conducted performance engineering for BBVA's e-commerce system, reducing response times by 80%.

We enhanced Singularity University's digital journey with specialized web and mobile testing.

Our experts developed scalable, resilient frameworks that ensured high-performance native apps.

We partnered with Threads to launch a high-quality communication platform from product development to delivery.

We amplified the streaming experience for 85,000 concurrent viewers in Asia.

We optimized Pronto+'s software development processes with automated performance testing.

AIA benefited from our optimized testing, improved platform stability, and quality recommendations.
GeneXus accelerated its software cycle with GXtest, our specialized automation tool that halved regression testing time.

We helped Marine Credit Union to strengthen their system's robustness and prepared them for self-sustained maintenance.
We supported and empowered GO’s engineering team with the design and implementation of a mobile app delivery pipeline.

We Enhanced EsSalud's IT System for Peak Performance.

We assisted Keeeb in scaling up quality control efforts and accelerating its digital transformation.

We have helped ANEP-DGEIP to enhance its software development and quality through the implementation of test methodologies and DevOps strategies.

COVID-19: Collaborating to Develop Life-Saving Software.

We facilitated Mosca Hnos.' smooth transition to advanced systems while maintaining uninterrupted service.

We streamlined Macromercado's system transition with GeneXus platform testing.

We refined a banking application for a client with a rich history spanning over 150 years.

Our experts implemented a testing strategy from scratch in a new Scrum team to enhance their AI-based web and mobile apps.
What Others Say...
"Abstracta is an excellent Quality Assurance partner, focused, motivated and skilled within their area of expertise. They delivered as expected and proposed organizational and process improvements which makes them a good long-term partner. Excellent job, thanks!"

Johan Frentz
"We were very impressed with Abstracta’s contractor and she seemed like a very good fit for what we had been looking for this project. I also wanted to commend and thank you for the quality of requested."

Steve Reichgut
VP of Engineering Benefit Cosmetics
"Abstracta management understood our unique testing needs and matched us up with a talented engineer who was quick to get up to speed with Gatling and our test environment. He asked apt questions, gave excellent status reports, and was overall a pleasure to work with. We look forward to continuing to work with Abstracta as the need arises."

Melissa Chawla
SR Manager Performance Engineering Shutterfly
Ready to improve the quality of your software? Our experienced team has helped startups to Fortune 500 companies. Fill out the form below and let's get started!
or email us at [email protected]
What is Quality Assurance (QA) in Software Testing?

What Is Quality Assurance?
History of quality assurance, what’s an example of quality assurance in practice.
- 4.1 1. Ensuring Customer Satisfaction:
- 4.2 2. Minimizing Costs and Risks:
- 4.3 3. Enhancing Brand Reputation:
- 4.4 4. Compliance with Standards and Regulations:
- 4.5 5. Continuous Improvement:
- 4.6 6. Increased Efficiency and Productivity:
- 5.1 Test Planning and Strategy
- 5.2 Test Design and Execution
- 5.3 Defect Management
- 5.4 Configuration Management
- 5.5 Metrics and Reporting
What are the types of Quality Assurance Functions?
Quality assurance principles.
- 8.2 Inspections
- 8.3 Statistical Process Control (SPC)
- 8.4 Risk Analysis
- 8.5 Root Cause Analysis
- 9.1 SQA Management Plan
- 9.2 Set the Goals
- 9.3 Re-check and Multi-Test
- 9.4 Measure Change Impact
- 9.5 Manage and Improve Communication
- 10.1 1. Analyzing the requirements:
- 10.2 2. Planning tests:
- 10.3 3. Designing the tests:
- 10.4 4. Executing the tests and reporting defects (if any):
- 10.5 5. Running re-tests to ensure safety:
- 10.6 6. Running release tests:
Benefits of Software Quality Assurance
Disadvantage of sqa, elements of software quality assurance, how is quality assurance measured.
- 15.1 1. Develop a QA plan:
- 15.2 2. Define clear acceptance criteria:
- 15.3 3. Use a version control system:
- 15.4 4. Perform regular code reviews:
- 15.5 5. Use automated testing:
- 15.6 6. Use multiple testing environments:
- 15.7 7. Track and report defects:
- 15.8 8. Perform regression testing:
- 15.9 9. Continuous integration and delivery (CI/CD):
- 15.10 10. Monitor and improve:
Quality Assurance Checklist
Tools for effective software qa.
Quality Assurance (QA) in software testing is a crucial process that ensures software products meet the highest quality requirements, ensuring flawless performance and customer satisfaction. It involves planning, executing, and monitoring testing operations to identify faults, minimize risks, and optimize overall program quality. With QA as a guiding light, developers can confidently navigate the vast field of software development, ensuring their products excel in the competitive digital realm.
So wait no more and take your business to its next level with QA!
Table Of Contents
- 1 What Is Quality Assurance?
- 2 History of Quality Assurance
- 3 What’s An Example of Quality Assurance in Practice?
- 4.1 1. Ensuring Customer Satisfaction:
- 4.2 2. Minimizing Costs and Risks:
- 4.3 3. Enhancing Brand Reputation:
- 4.4 4. Compliance with Standards and Regulations:
- 4.5 5. Continuous Improvement:
- 4.6 6. Increased Efficiency and Productivity:
- 6 What are the types of Quality Assurance Functions?
- 7 Quality Assurance Principles
- 10.1 1. Analyzing the requirements:
- 10.2 2. Planning tests:
- 10.3 3. Designing the tests:
- 10.4 4. Executing the tests and reporting defects (if any):
- 10.5 5. Running re-tests to ensure safety:
- 10.6 6. Running release tests:
- 11 Benefits of Software Quality Assurance
- 12 Disadvantage of SQA
- 13 Elements of Software Quality Assurance
- 14 How is Quality Assurance Measured?
- 16 Quality Assurance Checklist
- 17 Tools for Effective Software QA
- 18 Conclusion
Quality Assurance is the proactive process of ensuring that products, services, or processes meet or exceed predefined quality standards. It involves a systematic approach to prevent defects, errors, or inconsistencies throughout the development or production lifecycle. QA is like having a vigilant guardian ensuring everything is in place to deliver a product or service that consistently meets the highest quality, reliability, and customer satisfaction levels.
How about we take a trip back to ancient times to explore the history of Quality Assurance ?
The history of quality assurance can be traced back centuries, with early examples found in ancient civilizations like Egypt and Greece. Can you believe that? However, it was in the Middle Ages that formal systems emerged to ensure quality.
Key milestones in the history of quality assurance include:
- The development of interchangeable parts by Eli Whitney in the late 18th century. This innovation made it possible to mass-produce complex machines with greater efficiency and accuracy.
- The introduction of statistical process control (SPC) by Walter Shewhart in the early 20th century. SPC uses statistical tools to monitor and control production processes, identifying and eliminating sources of variation.
- The rise of Total Quality Management (TQM) in the mid-20th century. TQM emphasizes continuous improvement in all aspects of an organization, including quality, design, and customer service.
- The development of the International Organization for Standardization (ISO) 9000 series of quality management standards in the 1980s. ISO 9000 provides a framework for organizations to implement quality management systems.
Today, quality assurance is a key function in all industries, from manufacturing and software development to healthcare and education. It plays a vital role in ensuring that products and services meet customer expectations and regulatory requirements.
Let’s imagine a company that manufactures mobile phones. Here’s how they might use quality assurance:
- Review the design specifications to ensure they meet customer requirements.
- Conduct usability testing to ensure the phone is easy to use.
- Test the software for bugs and defects.
- Verify that the phone meets all performance requirements.
- Conduct stress testing to ensure the phone can withstand normal wear and tear.
- Inspect finished products for any defects.
- Conduct random testing to ensure quality standards are being met.
- Monitor customer feedback and complaints.
- Issue recalls or software updates if necessary.
Here are some more examples of quality assurance in practice:
In software development:
- Testing software to ensure it is free of bugs and defects.
- Writing clear and concise documentation that users can understand.
- Conducting user testing to ensure that the software is user-friendly.
In manufacturing:
- Inspecting products to ensure they meet quality standards.
- Testing materials to ensure they are safe and durable.
- Maintaining a clean and organized production environment.
In healthcare:
- Administering medications and treatments correctly.
- Monitoring patients for signs of complications.
- Maintaining a clean and sterile environment.
In education:
- Developing a curriculum that meets educational standards.
- Providing students with access to qualified teachers and resources.
- Assessing student learning to ensure they are meeting expectations.
These are just a few examples of how quality assurance is used in different industries. The specific techniques and tools will vary depending on the organization and the product or service being produced.
QA Used by the Industry
Different industries use different approaches to quality assurance, but the basic principles are the same. Here are some examples:
- Software Development: Unit testing, integration testing, system testing, acceptance testing.
- Manufacturing: Quality control inspections, statistical process control, defect tracking.
- Construction: Building inspections, code compliance checks, material testing.
- Healthcare: Medical record review, quality of care audits, infection control measures.
Importance of Quality Assurance
Quality assurance plays a vital role in various industries and sectors. Here are a few key reasons why it is of paramount importance:
1. Ensuring Customer Satisfaction:
Quality assurance is the backbone of delivering products or services that meet or exceed customer expectations. By systematically checking and verifying quality standards, organizations can instill customer confidence, build trust, and establish long-term relationships.
2. Minimizing Costs and Risks:
Implementing effective quality assurance processes helps identify and rectify defects or issues early on, reducing the costs associated with rework, product recalls, or customer complaints. Organizations can save time and resources by proactively mitigating risks and addressing quality concerns.
3. Enhancing Brand Reputation:
A strong focus on quality assurance translates into a reputable brand image. Consistently delivering high-quality products or services builds a positive reputation in the market, attracting more customers and fostering loyalty. A solid brand reputation is invaluable for long-term success and competitiveness.
4. Compliance with Standards and Regulations:
Many industries have strict regulations and quality standards that must be met. Quality assurance ensures adherence to these requirements, enabling organizations to comply with legal and industry-specific guidelines. Non-compliance can lead to penalties, legal issues, or damage to the organization’s reputation.
5. Continuous Improvement:
It fosters an organization’s continuous improvement culture. By monitoring processes, collecting data, and analyzing performance metrics, organizations can identify areas for enhancement and implement corrective actions.
6. Increased Efficiency and Productivity:
Implementing QA practices streamlines workflows, identifies bottlenecks, and eliminates inefficiencies. Organizations can enhance productivity, reduce time-to-market, and gain a competitive edge by optimizing processes and reducing waste.
In conclusion, quality assurance is not just a checkbox on a to-do list but a fundamental aspect of any successful organization. By prioritizing QA, businesses can deliver products and services that consistently meet customer expectations, minimize risks and costs, enhance their reputation, comply with regulations, drive continuous improvement, and achieve excellence.
What are Software Quality Assurance Components?
Software Quality Assurance encompasses several key components that work together to ensure the delivery of high-quality software products. These components include:
Test Planning and Strategy
Test planning and strategy involve creating a comprehensive roadmap for testing activities. It includes defining test objectives, identifying test deliverables, determining test approaches, and allocating resources. Planning and strategizing set the foundation for efficient and effective testing, ensuring that the right tests are conducted at the right time.
Test Design and Execution
Test design and execution involve creating and executing test cases for the software. This component focuses on designing test scenarios and creating detailed test cases that cover various aspects of the software’s functionality. Test execution involves running these test cases, capturing results, and verifying that the software behaves as expected.
Defect Management
Defect management is identifying, documenting, and tracking software defects or issues. It involves capturing detailed information about each defect, prioritizing them based on severity, and facilitating resolution. Defect management ensures that identified issues are properly addressed, allowing for the delivery of a high-quality, bug-free software product.
Configuration Management
Configuration management manages and controls software configuration elements changes throughout the development lifecycle. It includes version control, change management, and guaranteeing the software’s integrity and consistency. It helps maintain a stable and regulated environment, reducing risks associated with incorrect or unauthorized modifications.
Metrics and Reporting
Metrics and reporting involve gathering and analyzing important data during testing to draw useful insights. These metrics include test coverage, defect density, test execution progress, and other performance metrics. They provide visibility into the software’s quality, allowing for informed decision-making and continual development.
Quality assurance functions can vary depending on the industry and organizational context. However, some common types of functions include:
1. Product QA: This role is responsible for ensuring the quality of the final product or service by setting procedures, standards, and controls to meet quality goals and meet client expectations.
2. Process QA: Process quality assurance is creating and implementing process standards, performing audits, and identifying areas for improvement to maximize efficiency and quality.
3. Compliance QA: Compliance quality assurance ensures that legal, regulatory, and industry-specific requirements are followed to prevent penalties, legal challenges, and reputational harm.
4. Supplier QA: Supplier quality assurance manages the quality of goods or services delivered by third-party vendors by evaluating their capabilities, conducting audits, and creating quality agreements.
5. Customer QA: Customer quality assurance involves gathering client input, conducting customer satisfaction surveys, and taking action to improve customer experience and satisfaction. This includes gathering input, conducting surveys, and taking action to improve customer experience.
Quality assurance is guided by several key principles that form the foundation for effective quality management . These principles include:
1. Customer focus is essential for quality assurance, as it involves understanding the client’s needs, expectations, and satisfaction. Organizations can ensure that their goods or services meet customer expectations by putting the customer at the center of their quality initiatives.
2. It involves defining and optimizing processes , identifying process indicators, and monitoring and upgrading them to improve overall quality and efficiency.
3. It is committed to continuous improvement , which involves identifying areas for improvement and adopting corrective measures to generate continuous development, innovation, and excellence.
4. Data-driven decision-making involves gathering and analyzing relevant data, metrics, and performance indicators to assess the efficacy of quality procedures, detect trends, and promote evidence-based decision-making.
5. Quality assurance places a premium on empowering and engaging the team and cultivating a culture of ownership, responsibility, and continuous learning to promote quality improvement.
Quality Assurance Methods
To verify that products or services satisfy the intended quality standards, quality assurance incorporates a variety of methodologies and approaches. Here are some examples of common methods:
Audits are systematic examinations of processes, systems, or products to identify gaps, deviations, or noncompliance, allowing organizations to take remedial steps and improve overall quality and compliance.
Inspections
Inspection of work products or deliveries is essential to identify defects and prevent them from spreading and can be done at various stages of the development or production lifecycle.
Statistical Process Control (SPC)
Statistical Process Control monitors and controls processes using statistical methods to help organizations understand deviations, determine causes, and implement preventative or corrective steps to ensure process stability and consistency.
Risk Analysis
Risk analysis detects and analyzes possible product quality, performance, or dependability risks. It involves analyzing risks, evaluating their effect and likelihood, and implementing risk-mitigation methods to reduce the chance of quality-related difficulties.
Root Cause Analysis
Root Cause Analysis (RCA) is a problem-solving approach that involves studying symptoms, analyzing data, and determining the root causes of problems. It helps organizations identify root causes, implement remedial measures, and prevent similar situations from recurring.
Major Software Quality Assurance Activities
There are several activities of SQA that you must know about:
SQA Management Plan
This is usually the first step which involves planning your SQA approach and activities. List the activities included in your SQA and check with your team about their skills before proceeding.
Set the Goals
You have a plan and a team; now, you need to define the goals to achieve with your SQA. This will help you evaluate the project’s performance and assess your project’s progress.
Re-check and Multi-Test
Never depend on just one round of testing or one type of testing. Apply various testing approaches to ensure no area of the project is missed from testing.
Measure Change Impact
Resolving bugs sometimes creates other issues in the product. This is when you need to measure the change impact on your project. Perform a thorough inspection before and after the change to see if any other application area shows defects.
Manage and Improve Communication
The idea of SQA is to improve the collaboration between developers and testers. Keep everyone working on the project in the loop about what is happening and the next steps.
How to Implement Quality Assurance?
Implementing a software quality assurance plan is crucial to software development as it assists you in delivering the right product to the clients. Following are a few steps that can be implemented to bring out the best QA plan:
If you are interested in this software quality assurance plan, use Quality Assurance (QA) Training and join the course to improve your skills.

1. Analyzing the requirements:
Setting the requirements is a must, but knowing the need and result of it is necessary for any organization. Quality assurance professionals should be recruited to analyze various software requirements.
2. Planning tests:
Once the software is in line and the requirements have been analyzed, the second phase plans tests for the examination. The following areas are taken care of while planning tests:
The project’s budget, the strategy for testing the software, the span of testing, the deadlines, the methods used for testing, tools with which bugs will be tracked, etc.
3. Designing the tests:
After the planning stage comes the design stage of the test. The quality assurance team has to design some test cases that take care of the requirements of the software. Designing a test involves data, conditions, cases, and steps to verify each step of the program.
4. Executing the tests and reporting defects (if any):
After the test has been prepared, it is time to run it. The developers execute the test at the unit level. The quality assurance team does the execution of the trial at the UI and API levels.
Suppose a bug is encountered during the testing period. In that case, it is duly submitted to a tracking system for effective resolution.
5. Running re-tests to ensure safety:
After eliminating and fixing all the bugs, the quality assurance team re-runs the test to ensure no error has been left unchecked. The QA teams also run regression tests. Regression checks help to identify whether the bug fixing has made any changes in the existing functions.
6. Running release tests:
Once everything is done and the quality is assured, the developer team releases a notification stating all the implemented features, bug fixes, issues encountered, and the limitations of the software – for the release. Now, the quality assurance team modifies the test to match the software’s requirements of the latest developments – according to what the release will have.
There are some of the benefits of SQA:
- SQA produces high-quality software in less time, resources, and cost.
- SQA lays down a proper plan to conduct the testing process and improves intra-team and cross-team communication.
- It highlights the defects after every development stage rather than waiting till the end of SDLC.
- With SQA, you get high-quality products that increase the company’s market share.
- It cuts down the cost of maintenance as SQA gets product release right the first time. You can start working on the next project and only get back to the first one when required.
- SQA enhances product security by detecting issues in separate components of the product and fixing them before moving on to other areas.
Some of the disadvantages of SQA are:
- It can incur high costs, including deploying testing tools and hiring professional services, although this investment is nothing compared to the quality result you receive.
- It can be difficult to implement because SQA requires proper planning, designing, and implementation, which might be difficult for everyone on the team to understand.
There are 10 elements of SQA for you to follow:
- Software engineering standards: SQA teams need to give extra attention to adhering to the standards of software engineering teams.
- Technical reviews and audits: Verifying and validating the reviewing and auditing techniques at every stage of SDLC.
- Testing for quality control: Executing software testing to identify bugs in the application.
- Error collection and analysis: Recording the defects and analyzing their behavior and resolution.
- Metrics and measurement: SQA team employs multiple checks and measures to collect information about the product’s effectiveness and quality.
- Change management: Actively monitor the new changes and ensure that it does not induce defects in the existing functions of the software.
- Vendor management: Establish contacts with vendors to achieve collaborative success.
- Safety/security management: SQA highlights the security/safety vulnerabilities in the software.
- Risk management: The SQA heads the risk identification, analysis, and mitigation to assist in informed decision-making.
- Education: Stay updated on the recent trends, tools, standards, and market conditions.
There are many ways to measure the effectiveness of your quality assurance efforts. Here are a few common metrics:
- Defect rate: The number of defects found per unit of product or service.
- Customer satisfaction: How satisfied are your customers with the quality of your products or services?
- Return rate: The number of products or services returned due to defects.
- Warranty claims: The number of warranty claims against your products or services.
You can identify areas to improve your quality assurance efforts by tracking these metrics.
Best Practices – How do we Ensure Complete Quality Assurance?
To ensure that your software quality assurance (QA) verifies your software product and ensures it is fit for its intended purpose, Here are some best practices for implementing a QA process:
1. Develop a QA plan:
This should outline the scope of the QA process, the resources required, the roles and responsibilities of the QA team, and the testing schedule.
2. Define clear acceptance criteria:
These are the standards a software product must meet to be accepted by the client or end user. The acceptance criteria should be specific, measurable, attainable, relevant, and time-bound (SMART).
3. Use a version control system:
This allows multiple developers to work on the same codebase simultaneously and track changes made to the code.
4. Perform regular code reviews:
Code reviews help identify and fix problems early in development. Having at least two developers review the code before it is merged into the main codebase is best.
5. Use automated testing :
Automated testing can help reduce the time and effort required to perform testing. It is beneficial for regression testing, where the same tests are run repeatedly to ensure that changes to the code do not introduce new defects.
6. Use multiple testing environments:
Testing should be performed in settings representative of the intended production environment. This can help identify problems that may not be apparent in a development environment.
7. Track and report defects:
Use a system to record and track defects found during testing. This helps prioritize and address defects promptly.
8. Perform regression testing:
Regression testing helps ensure that changes made to the code do not introduce new defects or break existing functionality.
9. Continuous integration and delivery (CI/CD):
CICD entails integrating code changes into the main codebase and automatically building, testing, and deploying the software. This helps ensure that changes are integrated and deployed quickly and efficiently.
10. Monitor and improve:
Regularly monitor the QA process and gather feedback from stakeholders to identify areas for improvement. This can help continuously improve the quality of the software.
Creating an outstanding QA plan and test case management strategy involves proper documentation. The plan must be detailed and encompass all significant aspects, such as document plans, risk reports, mitigation, etc.
Here’s an essential checklist to get you started:
- Define your requirements. What are you trying to achieve with quality assurance?
- Identify your stakeholders. Who needs to be involved in the quality assurance process?
- Develop a quality plan. This will outline your quality assurance procedures and standards.
- Implement your quality plan. This may involve training employees, setting up a quality management system, and conducting inspections.
Monitor and analyze results. Track your progress and make adjustments to your quality plan as needed.
1. Performance testing tools – These tools are used to test the reliability and scalability of a product because every product has a crucial point where its performance goes down. Ex – Load Runner
2. Functional testing tools are used for error detection and giving it to the development team. Ex – Testsigma , Selenium and Cucumber
3. API testing tools – Used for testing the service layers or API. Ex – Postman
4. Unit testing tools – Used for testing individual codes(unit-wise). Ex- Code Coverage, SonarQube
5. Multi-browser testing tools – Used for cross-browser testing . Ex- Browser Stack
6. Test management tools – This helps you list all the requirements and write appropriate test cases. Ex – Mantis Bug Tracker and TestCollab
Finally, quality assurance is critical for guaranteeing that products or services satisfy the necessary quality standards. Organizations may increase customer happiness, reduce risks and costs, develop a credible brand image, and drive continuous improvement by employing efficient quality assurance practices. By embracing QA concepts and employing relevant methodologies, businesses may attain excellence and offer high-quality results that match consumer expectations.
In terms of SQA, Testsigma is a unified platform for streamlining testing operations.
Testsigma enables QA teams to simplify and optimize their testing operations with its straightforward UI, powerful features, and efficient test management capabilities. QA professionals can write, perform, and manage tests with Testsigma’s user-friendly platform, allowing them to focus on providing higher software quality while saving time and effort.
Frequently Asked Questions
What is the purpose of Quality Assurance?
What is the difference between QA and QC?
What is QA testing?
Case Studies

- Test consulting & tools assessment partnership providing test assessment approach along with its in-house tools evaluation frameworks across various areas ❶
- Test advisory and managed testing of their new banking systems and integration testing for the complex touchpoints for multiple Fiserv products ❷
- Functional testing for MIFID II compliance across various systems including Charles River, Black Rock Alladin and Unavista. Set up a team of domain experts in quick time and help client complete the testing as per the regulatory deadline ❸
- Functional, usability and performance testing of new digital wallet services for quick financial transactions, and performed end-to-end testing of mobile app and APIs ❹

- Test partnership across all digital and backend applications for functional testing and tst automation. Setting up the QA processes, automation solution and framework from scratch and defined the automation roadmap ❶
- Functional and regression testing for existing ERP and web application. Development of automation framework and script to run regression suite. Refined overall testing process and implemented agile processes for both SDLC & STLC ❷
- Designed test strategy and performed functional and system integration tests across the application & UAT support was provided for supply chain, stores sales, and warehouses system ❸
Health Care

- Test partnership for automation, functional & performance testing services in a hybrid model across their information management system tested on both functionality and database end ❶
- End-to-end functional, security and performance testing of solution for defects in the application before going live. Setting up QA/ testing processes and creating re-usable test cases, etc. ❷
- Created performance test scenarios & automated critical test cases for strategy style maintenance & batch execution of automation scripts for desktop-based application ❸
- Performed VAPT on the web application, web services, AWS S3 buckets and medical device as per OWASP guidelines & SANS Top 25. Created HIPAA compliance policy manual for client & assisted with its readiness assessment ❹

- End-to-end functional testing partnership and provided agile coaching and streamlined their test processes. Supported system, data migration, and performance testing for ongoing sprints & support on regression, exploratory, integration, and automation testing ❶
- Test advisory, end-to-end functional testing, test automation, implement integrated automated testing framework, test optimization techniques & helped in setting up CI/CD pipeline. Streamlined IT processes and artifacts, optimized resource skills, tool metrics & provided well-defined quality management and monitoring mechanism ❷
- End-to-end validations on their IoT system. Performed component level and system-level tests using JMeter, Grafana, and Prometheus integration was implemented for component level and system-level performance, scalability, stress and endurance tests ❸
Media & Publishing

- End-to-end test testing function operating in an onsite/ offshore model. Services included test automation and performance testing. Reduced regression cycle time using automation first approach and helped identify the application bottleneck ❶
- Test automation partnership for their money control application based on web, desktop and mobile (both Android & iOS). Leveraged its in-house test automation framework Tx-Automate built on the BDD approach to help create an end to end CI/ CD testing framework integrated with client’s in-house DevOps tools ❷
- Performed integration, functional, usability & regression testing the application & customized QA process as per client needs to implement enforced standards and to build consistency across the teams ❸
- Conducted POC to identify a suitable performance testing tool for the application & recommended HP load runner, perform tool and using these testing was performed of a complex application ❹
ISV & Telecom

- Staffing & managing of QA testing team to support a host of different applications. Implemented matured agile transformation QA practice and extensive experience of test and lifecycle automation helping to build the right QA capabilities ❶
- Device testing for their mobile application based on iOS and android platforms over different physical devices. Determined the transparent data transmission in the devices connected via HFP ❷
- Test automation partnership for client’s software used for client lifecycle management processes. Leveraged Tx-Automate to enhance the solution and build a team to rapidly scale test automation for their key financial services clients ❸

- Test partnership for cloud transformation initiative including moving applications portfolio to Cloud. Created detailed cloud migration testing strategy, test planning & execution ❶
- Crafted a compelling solution with its comprehensive managed testing centre of excellence (TCoE) for QA partnership, provided in a global delivery model (GDM) ❷
- Managed testing services including test advisory, functional testing, automation & performance testing for a critical initiative to move 35+ applications to cloud. Set up automation framework using in-house automation framework ‘Tx-Automate’ ❸
- Examined the current QA processes, people and tools, and benchmark against the industry best practices like TMMi/ CMMi. Provided futuristic QA and automation roadmap & helped them in standardizing the QA /testing process at organization level ❹
Related Blog

Robotic Process Automation (RPA): A Revolution in the Software Testing Industry

The Critical Need for Automation Testing in the Software Ecosystem
Subscribe Our Newsletter
Lorem ipsum dolor sit amet, consectetur
Opt-In. Read T&C
Request For Download
Read more on our Privacy Policy .
During your visit on our website, we collect personal information including but not limited to name, email address, contact number, etc. TestingXperts will collect and use your personal information for marketing, discussing the service offerings and provisioning the services you request. By clicking on the check box you are providing your consent on the same. In the future, if you wish to unsubscribe to our emails, you may indicate your preference by clicking on the “Unsubscribe” link in the email. Read more on our Privacy Policy
- Software Engineering Tutorial
- Software Development Life Cycle
- Waterfall Model
- Software Requirements
- Software Measurement and Metrics
- Software Design Process
- System configuration management
- Software Maintenance
- Software Development Tutorial
- Software Testing Tutorial
- Product Management Tutorial
- Project Management Tutorial
- Agile Methodology
- Selenium Basics
Software Quality Assurance – Software Engineering
Software Quality Assurance (SQA) is simply a way to assure quality in the software. It is the set of activities that ensure processes, procedures as well as standards are suitable for the project and implemented correctly.
Software Quality Assurance is a process that works parallel to Software Development. It focuses on improving the process of development of software so that problems can be prevented before they become major issues. Software Quality Assurance is a kind of Umbrella activity that is applied throughout the software process.
Table of Content
What is quality?
- Software Quality Assurance (SQA) encompasses
Elements of Software Quality Assurance (SQA)
Software quality assurance (sqa) focuses, software quality assurance (sqa) include, major software quality assurance (sqa) activities, benefits of software quality assurance (sqa), disadvantage of software quality assurance (sqa), frequently asked questions on software quality assurance (sqa).
Generally, the quality of the software is verified by third-party organizations like international standard organizations .
Quality in a product or service can be defined by several measurable characteristics. Each of these characteristics plays a crucial role in determining the overall quality.

Software Quality Assurance (SQA) encompasse s
SQA process Specific quality assurance and quality control tasks (including technical reviews and a multitiered testing strategy) Effective software engineering practice (methods and tools) Control of all software work products and the changes made to them a procedure to ensure compliance with software development standards (when applicable) measurement and reporting mechanisms
- Standards: The IEEE, ISO, and other standards organizations have produced a broad array of software engineering standards and related documents. The job of SQA is to ensure that standards that have been adopted are followed and that all work products conform to them.
- Reviews and audits: Technical reviews are a quality control activity performed by software engineers for software engineers. Their intent is to uncover errors. Audits are a type of review performed by SQA personnel (people employed in an organization) with the intent of ensuring that quality guidelines are being followed for software engineering work.
- Testing: Software testing is a quality control function that has one primary goal—to find errors. The job of SQA is to ensure that testing is properly planned and efficiently conducted for primary goal of software.
- Error/defect collection and analysis : SQA collects and analyzes error and defect data to better understand how errors are introduced and what software engineering activities are best suited to eliminating them.
- Change management: SQA ensures that adequate change management practices have been instituted.
- Education: Every software organization wants to improve its software engineering practices. A key contributor to improvement is education of software engineers, their managers, and other stakeholders. The SQA organization takes the lead in software process improvement which is key proponent and sponsor of educational programs.
- Security management: SQA ensures that appropriate process and technology are used to achieve software security.
- Safety: SQA may be responsible for assessing the impact of software failure and for initiating those steps required to reduce risk.
- Risk management : The SQA organization ensures that risk management activities are properly conducted and that risk-related contingency plans have been established.
The Software Quality Assurance (SQA) focuses on the following
-focuses.webp)
Software Quality Assurance (SQA)
- Software’s portability: Software’s portability refers to its ability to be easily transferred or adapted to different environments or platforms without needing significant modifications. This ensures that the software can run efficiently across various systems, enhancing its accessibility and flexibility.
- software’s usability: Usability of software refers to how easy and intuitive it is for users to interact with and navigate through the application. A high level of usability ensures that users can effectively accomplish their tasks with minimal confusion or frustration, leading to a positive user experience.
- software’s reusability: Reusability in software development involves designing components or modules that can be reused in multiple parts of the software or in different projects. This promotes efficiency and reduces development time by eliminating the need to reinvent the wheel for similar functionalities, enhancing productivity and maintainability.
- software’s correctness: Correctness of software refers to its ability to produce the desired results under specific conditions or inputs. Correct software behaves as expected without errors or unexpected behaviors, meeting the requirements and specifications defined for its functionality.
- software’s maintainability: Maintainability of software refers to how easily it can be modified, updated, or extended over time. Well-maintained software is structured and documented in a way that allows developers to make changes efficiently without introducing errors or compromising its stability.
- software’s error control: Error control in software involves implementing mechanisms to detect, handle, and recover from errors or unexpected situations gracefully. Effective error control ensures that the software remains robust and reliable, minimizing disruptions to users and providing a smoother experience overall.
- A quality management approach.
- Formal technical reviews.
- Multi testing strategy.
- Effective software engineering technology.
- Measurement and reporting mechanism.
- SQA Management Plan: Make a plan for how you will carry out the SQA throughout the project. Think about which set of software engineering activities are the best for project. check level of SQA team skills.
- Set The Check Points: SQA team should set checkpoints. Evaluate the performance of the project on the basis of collected data on different check points.
- Measure Change Impact: The changes for making the correction of an error sometimes re introduces more errors keep the measure of impact of change on project. Reset the new change to check the compatibility of this fix with whole project.
- Multi testing Strategy: Do not depend on a single testing approach. When you have a lot of testing approaches available use them.
- Manage Good Relations: In the working environment managing good relations with other teams involved in the project development is mandatory. Bad relation of SQA team with programmers team will impact directly and badly on project. Don’t play politics.
- Maintaining records and reports: Comprehensively document and share all QA records, including test cases, defects, changes, and cycles, for stakeholder awareness and future reference.
- Reviews software engineering activities: The SQA group identifies and documents the processes. The group also verifies the correctness of software product.
- Formalize deviation handling: Track and document software deviations meticulously. Follow established procedures for handling variances.
- SQA produces high quality software.
- High quality application saves time and cost.
- SQA is beneficial for better reliability.
- SQA is beneficial in the condition of no maintenance for a long time.
- High quality commercial software increase market share of company.
- Improving the process of creating software.
- Improves the quality of the software.
- It cuts maintenance costs. Get the release right the first time, and your company can forget about it and move on to the next big thing. Release a product with chronic issues, and your business bogs down in a costly, time-consuming, never-ending cycle of repairs.
There are a number of disadvantages of quality assurance.
- Cost: Some of them include adding more resources, which cause the more budget its not, Addition of more resources For betterment of the product.
- Time Consuming: Testing and Deployment of the project taking more time which cause delay in the project.
- Overhead : SQA processes can introduce administrative overhead, requiring documentation, reporting, and tracking of quality metrics. This additional administrative burden can sometimes outweigh the benefits, especially for smaller projects.
- Resource Intensive : SQA requires skilled personnel with expertise in testing methodologies, tools, and quality assurance practices. Acquiring and retaining such talent can be challenging and expensive.
- Resistance to Change : Some team members may resist the implementation of SQA processes, viewing them as bureaucratic or unnecessary. This resistance can hinder the adoption and effectiveness of quality assurance practices within an organization.
- Not Foolproof : Despite thorough testing and quality assurance efforts, software can still contain defects or vulnerabilities. SQA cannot guarantee the elimination of all bugs or issues in software products.
- Complexity : SQA processes can be complex, especially in large-scale projects with multiple stakeholders, dependencies, and integration points. Managing the complexity of quality assurance activities requires careful planning and coordination.
Software Quality Assurance (SQA) maintain a most important role in the ensuring the quality, reliability and efficiency of the product. By implementation of these control process which cause the improvement of the software engineering process . SQA gives a higher quality product which help to meet user expectations, its having some drawback also like Cost, time-consuming process, after maintaining the process of the SQA its improved the reliability and maintain the maintenance cost which affect in a future.
Overall, Software Quality Assurance (SQA) is important for the success in the project development in Software Engineering
What Does Do Software Quality Assurance (SQA) in software Development?
SQA makes sure that the software is made according to the need and checking its build.
How does Software Quality Assurance (SQA) help software work better?
SQA Finds the faults in the Software before its use, it will help to make software more trustable.
What parts are important in Software Quality Assurance (SQA)?
SQA Checks the software follows rules, it will learn from example, manage changes, check working well, educate teams, ensure security, and handle the risk.
Please Login to comment...
Similar reads.
- Software Engineering
- Software Testing
Improve your Coding Skills with Practice
What kind of Experience do you want to share?

What Are QA Test Cases?

When getting started with any type of QA testing , it’s always helpful to have test cases. But what are QA test cases, and why are they important? Read on to learn how they work, and how to write test cases.
What Should A Test Case Contain?
Well written test cases should contain the following aspects:
- Title or ID (ie: “LOG-1”)
- An association with a section of the site/app (ie: “Login”)
- Notes on any setup needed (for example, “access to a staging build” or “admin credentials”)
- Steps to follow to perform the test
- Expected results
Why Are Test Cases Important?

Most importantly, having test cases that all team members can review ensures two crucial details:
- There are no gaps in test coverage . Let’s say that QA hasn’t been informed about a particular feature or requirement. By reviewing test cases, other team members can immediately determine if there’s missing test coverage.
- Everyone is on the same page. Sometimes there are misunderstandings about how a feature is supposed to work. Any misconceptions can be identified faster and more easily when a developer or product manager can review test cases.
For more on creating test cases without team collaboration, see our article How to Write Test Cases Without Requirements .
Test Cases For Manual Testing
Manual testing is essential for the quality of a mobile app or website. In the beginning of the software development life cycle , manual QA testers often interact with the product owner and project manager, and collaborate with developers . Accordingly, this teamwork ensures that the product is developed with the best possible user experience . The result of that communication is manifested in the test cases. This is part of why test cases are invaluable for Engineering.

Now imagine if the above scenario happened and Shawn Carter had well-defined test cases. These would include all of the necessary setup information, steps to follow, and expected results. You could breathe a sigh of relief knowing that the sprint could go on. Test cases can be the difference between smooth sailing and total disaster.
(Teams can also minimize concerns with a QA lead being out by doing collaborative QA job sharing .)
Test Cases for Automation

Remember how manual QA collaborated with product managers and developers earlier on? The benefits of all that research make writing automated tests faster, easier, and without a need for going back to square one. Clarifying questions may come up, but redundancy will be decreased big-time. The result? A functional automated test suite that lets manual testers focus on new features — instead of spending days doing regression testing .
For more, see our guide to the Top 5 Test Cases to Automate .
Sample Test Cases and Template for a Login Page
Let’s get to the good stuff! Here are some sample test cases, using a template for a login page or screen. These test cases aren’t exhaustive. But they’re good simple examples of what test cases look like.
(If you’re viewing on a mobile device, we recommend using landscape mode.)
| ID | Requirement | Setup | Steps | Expected Result |
|---|---|---|---|---|
| LOG-1 | Login/Sign up menu button should function | Access to staging iOS/Android builds + cell service or internet connection | 1. Open app 2. Tap hamburger menu icon in top left 3. Tap "Login/Sign Up" button | User should be directed to login/sign up screen within 2 seconds. |
| LOG-2 | When logged in, tapping "Logout" in left nav menu should log the user out | Access to staging iOS/Android builds, cell service or internet connection, and valid account credentials | 1. Open app 2. Tap hamburger menu icon in top left 3. Tap "Login/Sign Up" button 4. Log in using valid account credentials 5. Tap hamburger menu icon in top left 6. Tap "Logout" | User should be logged out, and directed to home screen within 2 seconds. |
| LOG-3 | If user enters incorrect password, relevant error message should appear | Access to staging iOS/Android builds + cell service or internet connection | 1. Open app 2. Tap hamburger menu icon in top left 3. Tap "Login/Sign Up" button 4. Enter an email in "Email" field 5. Enter a password that is not valid 6. Tap "Login" button | Relevant error message should appear within 2 seconds, informing the user that the password was not valid. |
| LOG-4 | When user enters valid email address and password and taps Login, they should be logged in and taken to Home screen | Access to staging iOS/Android builds, cell service or internet connection, and valid account credentials | 1. Open app 2. Tap hamburger menu icon in top left 3. Tap "Login/Sign Up" button 4. Log in using valid account credentials | User should be directed to home screen as a logged in user within 2 seconds. |
| LOG-5 | Email address field should not be case sensitive | Access to staging iOS/Android builds, cell service or internet connection, and valid account credentials | 1. Open app 2. Tap hamburger menu icon in top left 3. Tap "Login/Sign Up" button 4. In the email field, enter a valid registered email address, but use capital letters for some of the characters (for example, [email protected]) 5. Enter valid password 6. Tap "Login" | User should be directed to home screen as a logged in user within 2 seconds. |
| LOG-6 | Password field should be case sensitive | Access to staging iOS/Android builds + cell service or internet connection | 1. Open app 2. Tap hamburger menu icon in top left 3. Tap "Login/Sign Up" button 4. Enter an email in "Email" field 5. Enter a password that would be valid, but use capital letters for characters that should have been lowercase, and lowercase letters for characters that should have been caps 6. Tap "Login" | Relevant error message should appear within 2 seconds, informing the user that the password was not valid. |
In conclusion, test cases are a great way to make sure that the mobile app or website has the test coverage it needs. For instance, test cases increase efficiency, and can even help automated testing. And while it’s always best to have seasoned QA engineers do the testing, test cases also allow backup QA to jump in and help out in a pinch.
Need Test Cases?
Looking for QA testers to write test cases for your app or site (or to test it)? Check out our QA testing services .
About the author

[…] Writing test cases […]
Leave a Reply Cancel reply
Your email address will not be published. Required fields are marked *
Making quality assurance smart
For decades, outside forces have dictated how pharmaceutical and medtech companies approach quality assurance. The most influential force remains regulatory requirements. Both individual interpretations of regulations and feedback received during regulatory inspections have shaped quality assurance systems and processes. At the same time, mergers and acquisitions, along with the proliferation of different IT solutions and quality software, have resulted in a diverse and complicated quality management system (QMS) landscape. Historically, the cost of consolidating and upgrading legacy IT systems has been prohibitively expensive. Further challenged by a scarcity of IT support, many quality teams have learned to rely on the processes and workflows provided by off-the-shelf software without questioning whether they actually fit their company’s needs and evolving regulatory requirements.
In recent years, however, several developments have enabled a better way. New digital and analytics technologies make it easier for quality teams to access data from different sources and in various formats, without replacing existing systems. Companies can now build dynamic user experiences in web applications at a fraction of the cost of traditional, enterprise desktop software; this development raises the prospect of more customized, user-friendly solutions. Moreover, regulators, such as the FDA, are increasingly focused on quality systems and process maturity. 1 MDIC Case for Quality program. The FDA also identified the enablement of innovative technologies as a strategic priority, thereby opening the door for constructive dialogue about potential changes. 2 Technology Modernization Action Plan, FDA.
Smart quality at a glance
“Smart quality” is a framework that pharma and medtech companies can apply to redesign key quality assurance processes and create value for the organization.
Smart quality has explicit objectives:
- to perceive and deliver on multifaceted and ever-changing customer needs
- to deploy user-friendly processes built organically into business workflows, reimagined with leading-edge technologies
- to leapfrog existing quality management systems with breakthrough innovation, naturally fulfilling the spirit—not just the letter—of the regulations
The new ways in which smart quality achieves its objectives can be categorized in five building blocks (exhibit).
To learn more about smart quality and how leading companies are reimagining the quality function, please see “ Smart quality: Reimagining the way quality works .”
The time has arrived for pharmaceutical and medtech companies to act boldly and reimagine the quality function. Through our work on large-scale quality transformation projects and our conversations with executives, we have developed a new approach we call “smart quality” (see sidebar, “Smart quality at a glance”). With this approach, companies can redesign key quality processes and enable design-thinking methodology (to make processes more efficient and user-friendly), automation and digitization (to deliver speed and transparency), and advanced analytics (to provide deep insights into process capability and product performance).
The quality assurance function thereby becomes a driver of value in the organization and a source of competitive advantage—improving patient safety and health outcomes while operating efficiently, effectively, and fully aligned with regulatory expectations. In our experience, companies applying smart quality principles to quality assurance can quickly generate returns that outweigh investments in new systems, including line-of-sight impact on profit; a 30 percent improvement in time to market; and a significant increase in manufacturing and supply chain reliability. Equally significant are improvements in customer satisfaction and employee engagement, along with reductions in compliance risk.
Revolutionizing quality assurance processes
The following four use cases illustrate how pharmaceutical and medtech companies can apply smart quality to transform core quality assurance processes—including complaints management, quality management review, deviations investigations, and supplier risk management, among others.
1. Complaint management
Responding swiftly and effectively to complaints is not only a compliance requirement but also a business necessity. Assessing and reacting to feedback from the market can have an immediate impact on patient safety and product performance. Today, a pharmaceutical or medtech company may believe it is handling complaints well if it has a single software deployed around the globe for complaint management, with some elements of automation (for example, flagging reportable malfunctions in medical devices) and several processing steps happening offshore (such as intake, triage, and regulatory reporting).
Yet, for most quality teams, the average investigation and closure cycle time hovers around 60 days—a few adverse events are reported late every month, and negative trends are addressed two or more months after the signals come in. It can take quality assurance teams even longer to identify complaints that collectively point to negative trends for a particular product or device. At the same time, less than 5 percent of incoming complaints are truly new events that have never been seen before. The remainder of complaints can usually be categorized into well-known issues, within expected limits; or previously investigated issues, in which root causes have been identified and are already being addressed.
The smart quality approach improves customer engagement and speed
By applying smart quality principles and the latest technologies, companies can reduce turnaround times and improve the customer experience. They can create an automated complaint management process that reduces costs yet applies the highest standards:
- For every complaint, the information required for a precise assessment is captured at intake, and the event is automatically categorized.
- High-risk issues are immediately escalated by the system, with autogenerated reports ready for submission.
- New types of complaints and out-of-trend problems are escalated and investigated quickly.
- Low-risk, known issues are automatically trended and closed if they are within expected limits or already being addressed.
- Customer responses and updates are automatically available.
- Trending reports are available in real time for any insights or analyses.
To transform the complaint management process, companies should start by defining a new process and ensuring it meets regulatory requirements. The foundation for the new process can lie in a structured event assessment that allows automated issue categorization based on the risk level defined in the company’s risk management documentation. A critical technological component is the automation of customer complaint intake; a dynamic front-end application can guide a customer through a series of questions (Exhibit 1). The application captures only information relevant to a specific complaint evaluation, investigation, and—if necessary—regulatory report. Real-time trending can quickly identify signals that indicate issues exceeding expected limits. In addition, companies can use machine learning to scan text and identify potential high-risk complaints. Finally, risk-tailored investigation pathways, automated reporting, and customer response solutions complete the smart quality process. Successful companies maintain robust procedures and documentation that clearly explain how the new process reliably meets specific regulatory requirements. Usually, a minimal viable product (MVP) for the new process can be built within two to four months for the first high-volume product family.
In our experience, companies that redesign the complaint management process can respond more swiftly—often within a few hours—to reduce patient risk and minimize the scale and impact of potential issues in the field. For example, one medtech company that adopted the new complaint management approach can now automatically assess all complaints and close more than 55 percent of them in 24 hours without human intervention. And few, if any, reportable events missed deadlines for submission. Now, subject matter experts are free to focus on investigating new or high-risk issues, understanding root causes, and developing the most effective corrective and preventive actions. The company also reports that its customers prefer digital interfaces to paper forms and are pleased to be updated promptly on their status and resolution of their complaints.
2. Quality management review
Real-time performance monitoring is crucial to executive decision making at pharmaceutical and medtech companies. During a 2019 McKinsey roundtable discussion, 62 percent of quality assurance executives rated it as a high priority for the company, exceeding all other options.
For many companies today, the quality review process involves significant manual data collection and chart creation. Often, performance metrics focus on quality compliance outcomes and quality systems—such as deviation cycle times—at the expense of leading indicators and connection to culture and cost. Managers and executives frequently find themselves engaged in lengthy discussions, trying to interpret individual metrics and often missing the big picture.
Although many existing QMS solutions offer automated data-pull and visualization features, the interpretation of complex metric systems and trends remains largely a manual process. A team may quickly address one performance metric or trend, only to learn several months later that the change negatively affected another metric.
The smart quality approach speeds up decision making and action
By applying smart quality principles and the latest digital technologies, companies can get a comprehensive view of quality management in real time. This approach to performance monitoring allows companies to do the following:
- automatically collect, analyze, and visualize relevant leading indicators and outcomes on a simple and intuitive dashboard
- quickly identify areas of potential risk and emerging trends, as well as review their underlying metrics and connections to different areas
- rapidly make decisions to address existing or emerging issues and monitor the results
- adjust metrics and targets to further improve performance as goals are achieved
- view the entire value chain and create transparency for all functions, not just quality
To transform the process, companies should start by reimagining the design of the process and settling on a set of metrics that balances leading and lagging indicators. A key technical enabler of the system is establishing an interconnected metrics structure that automates data pull and visualization and digitizes analysis and interpretation (Exhibit 2). Key business processes, such as regular quality management reviews, may require changes to include a wider range of functional stakeholders and to streamline the review cascade.
Healthcare companies can use smart quality to redesign the quality management review process and see results quickly. At one pharmaceutical and medtech company, smart visualization of connected, cross-functional metrics significantly improved the effectiveness and efficiency of quality management review at all levels. Functions throughout the organization reported feeling better positioned to ascertain the quality situation quickly, support decision making, and take necessary actions. Because of connected metrics, management can not only see alarming trends but also link them to other metrics and quickly align on targeted improvement actions. For example, during a quarterly quality management review, the executive team linked late regulatory reporting challenges to an increase in delayed complaint submissions in some geographic regions. Following the review, commercial leaders raised attention to this issue in their respective regions, and in less than three months, late regulatory reporting was reduced to zero. Although the company is still in the process of fully automating data collection, it has already noticed a significant shift in its work. The quality team no longer spends the majority of its time on data processing but has pivoted to understanding, interpreting, and addressing complex and interrelated trends to reduce risks associated with quality and compliance.
Healthcare companies can use smart quality to redesign the quality management review process and see results quickly.
3. Deviation or nonconformance investigations
Deviation or nonconformance management is a critical topic for companies today because unaddressed issues can lead to product recalls and reputational damage. More often, deviations or nonconformances can affect a company’s product-release process, capacity, and lead times. As many quality teams can attest, the most challenging and time-consuming part of a deviation or nonconformance investigation is often the root cause analysis. In the best of circumstances, investigators use a tracking and trending system to identify similar occurrences. However, more often than not, these systems lack good classification of root causes and similarities. The systems search can become another hurdle for quality teams, resulting in longer lead times and ineffective root cause assessment. Not meeting the standards defined by regulators regarding deviation or nonconformance categorization and root cause analysis is one of the main causes of warning letters or consent decrees.
The smart quality approach improves effectiveness and reduces lead times
Our research shows companies that use smart quality principles to revamp the investigation process may reap these benefits:
- all pertinent information related to processes and equipment is easily accessible in a continuously updated data lake
- self-learning algorithms predict the most likely root cause of new deviations, thereby automating the review of process data and statements
In our experience, advanced analytics is the linchpin of transforming the investigation process. The most successful companies start by building a real-time data model from local and global systems that continuously refreshes and improves the model over time. Natural language processing can generate additional classifications of deviations or nonconformances to improve the quality and accuracy of insights. Digitization ensures investigators can easily access graphical interfaces that are linked to all data sources. With these tools in place, companies can readily identify the most probable root cause for deviation or nonconformance and provide a fact base for the decision. Automation also frees quality assurance professionals to focus on corrective and preventive action (Exhibit 3).
Pharmaceutical and medtech companies that apply these innovative technologies and smart quality principles can see significant results. Our work with several companies shows that identifying, explaining, and eliminating the root causes of recurring deviations and nonconformances can reduce the overall volume of issues by 65 percent. Companies that use the data and models to determine which unexpected factors in processes and products influence the end quality are able to control for them, thereby achieving product and process mastery. What’s more, by predicting the most likely root causes and their underlying drivers, these companies can reduce the investigation cycle time for deviations and nonconformances by 90 percent.
4. Supplier quality risk management
Drug and medical device supply chains have become increasingly global, complex, and opaque as more pharmaceutical and medtech companies outsource major parts of production to suppliers and contract manufacturing organizations (CMOs). More recently, the introduction of new, complex modalities, such as cell therapy and gene editing, has further increased pressure to ensure the quality of supplier products. Against this backdrop, it is critical to have a robust supplier quality program that can proactively identify and mitigate supplier risks or vulnerabilities before they become material issues.
Today, many companies conduct supplier risk management manually and at one specific point in time, such as at the beginning of a contract or annually. Typically, risk assessments are done in silos across the organization; every function completes individual reports and rarely looks at supplier risk as a whole. Because the results are often rolled up and individual risk signals can become diluted, companies focus more on increasing controls than addressing underlying challenges.
The smart quality approach reduces quality issues and optimizes resources
Companies that break down silos and apply a more holistic risk lens across the organization have a better chance of proactively identifying supplier quality risks. With smart quality assurance, companies can do the following:
- identify vulnerabilities by utilizing advanced analytics on a holistic set of internal and external supplier and product data
- ensure real-time updates and reviews to signal improvements in supplier quality and any changes that may pose an additional risk
- optimize resource allocation and urgency of action, based on the importance and risk level of the supplier or CMO
Current technologies make it simpler than ever to automatically collect meaningful data. They also make it possible to analyze the data, identify risk signals, and present information in an actionable format. Internal and supplier data can include financials, productivity, and compliance metrics. Such information can be further enhanced by publicly available external sources—such as regulatory reporting, financial statements, and press releases—that provide additional insights into supplier quality risks. For example, using natural language processing to search the web for negative press releases is a simple yet powerful method to identify risks.
Would you like to learn more about our Life Sciences Practice ?
Once a company has identified quality risks, it must establish a robust process for managing these risks. Mitigation actions can include additional monitoring with digital tools, supporting the supplier to address the sources of issues, or deciding to switch to a different supplier. In our experience, companies that have a deep understanding of the level of quality risk, as well as the financial exposure, have an easier time identifying the appropriate mitigation action. Companies that identify risks and proactively mitigate them are less likely to experience potentially large supply disruptions or compliance findings.
Many pharmaceutical and medtech companies have taken steps to improve visibility into supplier quality risks by using smart quality principles. For example, a large pharmaceutical company that implemented this data-driven approach eliminated in less than two years major CMO and supplier findings that were identified during audits. In addition, during the COVID-19 pandemic, a global medtech company was able to proactively prevent supply chain disruptions by drawing on insights derived from smart quality supplier risk management.
Getting started
Pharmaceutical and medtech companies can approach quality assurance redesign in multiple ways. In our experience, starting with two or three processes, codifying the approach, and then rolling it out to more quality systems accelerates the overall transformation and time to value.
Smart quality assurance starts with clean-sheet design. By deploying modern design techniques, organizations can better understand user needs and overcome constraints. To define the solution space, we encourage companies to draw upon a range of potential process, IT, and analytics solutions from numerous industries. In cases where the new process is substantially different from the legacy process, we find it beneficial to engage regulators in an open dialogue and solicit their early feedback to support the future-state design.
Once we arrive at an MVP that includes digital and automation elements, companies can test and refine new solutions in targeted pilots. Throughout the process, we encourage companies to remain mindful of training and transition planning. Plans should include details on ensuring uninterrupted operations and maintaining compliance during the transition period.
The examples in this article are not exceptions. We believe that any quality assurance process can be significantly improved by applying a smart quality approach and the latest technologies. Pharmaceutical and medtech companies that are willing to make the organizational commitment to rethink quality assurance can significantly reduce quality risks, improve their speed and effectiveness in handling issues, and see long-term financial benefits.
Note: The insights and concepts presented here have not been validated or independently verified, and future results may differ materially from any statements of expectation, forecasts, or projections. Recipients are solely responsible for all of their decisions, use of these materials, and compliance with applicable laws, rules, and regulations. Consider seeking advice of legal and other relevant certified/licensed experts prior to taking any specific steps.
Explore a career with us
Related articles.

Smart quality: Reimagining the way quality works
Ready for launch: Reshaping pharma’s strategy in the next normal

Healthcare innovation: Building on gains made through the crisis

IMAGES
VIDEO
COMMENTS
Software Testing Client Project Case Study. We are often asked what software testing is. The video below shares a solid definition of the term. But we thought a software testing project case study might be helpful to better understand what software testers do on a typical day. This includes testing software, writing requirement documents for ...
Our engineer was also responsible for providing live or recorded demonstrations of completed tasks, as well as extensive documentation of the testing behavior. Test Cases Written (64) Implementation Time (3 weeks) Execution Time (30 minutes) Customer Satisfaction. The software quality assurance professionals at Beta Breakers test web and mobile ...
To ensure maximum productivity, all testers receive specialized education on their clients' industry and issues before starting projects. Learn more about how QA Consultants helps our customers with software testing and quality assurance by reading our case studies. QA Consultants is an award-winning provider of software testing and quality ...
1) Web applications. Consider a scenario where a software development company is building a web application for online banking. Here, QA plays a pivotal role in ensuring the application's security, functionality, and user experience. Here are some examples of QA practices for web application testing:
Software Quality Assurance Case Study. QA case study: Usually startups are less concerned about the quality and in many cases developers are doing QA part of work, we understand that it saves cost and gives chance to release product in a short period of time but on another hand clients will face poor quality this is exactly the situation we saw ...
c) Example 3: Government Digital Services Improvement . 2) Conclusion . Software Testing Case Studies . Exploring real-world examples of Software Testing can give detailed insights into best practices and common challenges. These case studies highlight how different organisations have approached Software Testing to ensure quality and reliability.
Get Started. Experience a Free 30 Min Consultation with an Expert. Schedule a Free Consultation. Read our software testing case studies to find what challenges our testing experts has faces and how they encountered them to deliver client satisfaction in various verticals like automation Testing , mobile testing, security testing and more.
Case studies Industry All AI AR Banking Betting Big Data Branding E-commerce E-learning Fintech Healthcare Interactive video platform Investment Management Logistics and Supply Chain Marketing Marketing & Branding Media & Entertainment Mobile Games NFT Office Software Online Meetings Procurement Project Management Software Development Sport ...
SQA, sometimes referred to as software quality assurance, is only a technique for ensuring the quality of the programme. It is a group of steps performed to ensure that the standards, procedures ...
Browse our library of software testing case studies to see how startups to Fortune 500s have partnered with Abstracta to improve their software quality. ... "Abstracta is an excellent Quality Assurance partner, focused, motivated and skilled within their area of expertise. They delivered as expected and proposed organizational and process ...
Conclusion. Quality Assurance (QA) in software testing is a crucial process that ensures software products meet the highest quality requirements, ensuring flawless performance and customer satisfaction. It involves planning, executing, and monitoring testing operations to identify faults, minimize risks, and optimize overall program quality.
Using Exploratory Data Analysis to Improve the Fresh Foods Ordering Process in Retail Stores. This case study presents a real-world example of how the thought processes of data scientists can contribute to quality practice. See how explorative data analysis and basic statistics helped a grocery chain reduce inefficiencies in its retail ...
Case Study: Quality Assurance Client Overview The client, Thomson Reuters, is the world's leading source of intelligent information for businesses and ... effectiveness of testing and, in effect, the software quality. The company needed to establish a quality assurance (QA) structure that would meet industry standards. Insigma's Solution ...
Setting up QA/ testing processes and creating re-usable test cases, etc. . Created performance test scenarios & automated critical test cases for strategy style maintenance & batch execution of automation scripts for desktop-based application . Performed VAPT on the web application, web services, AWS S3 buckets and medical device as per OWASP ...
You can improve the quality of the software and the user experience. A higher quality product means more satisfied customers and higher profits. Popular test case management tools. Test case management tools help you manage software and hardware development. These tools track your test cases, bugs, and other important information related to ...
Software Quality Assurance (SQA) is simply a way to assure quality in the software. It is the set of activities that ensure processes, procedures as well as standards are suitable for the project and implemented correctly. Software Quality Assurance is a process that works parallel to Software Development. It focuses on improving the process of ...
The book presents a comprehensive discussion on software quality issues and software quality assurance (SQA) principles and practices, and lays special emphasis on implementing and managing SQA. Primarily designed to serve three audiences; universities and college students, vocational training participants, and software engineers and software development managers, the book may be applicable to ...
In the beginning of the software development life cycle, manual QA testers often interact with the product owner and project manager, and collaborate with developers. Accordingly, this teamwork ensures that the product is developed with the best possible user experience. The result of that communication is manifested in the test cases.
Brennan, James, "Case Study: Implementing Tools for Software Quality Assurance" (2009). Regis University Student Publications (comprehensive collection). 794. This Thesis - Open Access is brought to you for free and open access by the Regis University Student Publications at ePublications at Regis University. It has been accepted for inclusion ...
Pentesting of 11,500 IPs and 50 Apps for a Warehouse Automation Provider. We share software testing and QA projects fulfilled for our customers from a range of domains, validating different aspects of software and employing both manual and automated efforts.
3 Quality Assurance Framework Figure 1. Quality Assurance Framework Figure 1 shows the integration of our quality assurance framework with the SOA of the performance management system. The OpenSTA client simulates user actions in the Enterprise PM Portal. There are log files that contain detailed information from each component of the system.
This paper reports on data collected in December of 2007 concerning the implementation of a Software Quality Assurance tool at a Fortune 500 company in August 2006. The data analysis reveals problems that can arise while applying a Software Quality Assurance tool, the means required to employ a quality process and how to resolve major issues ...
Case study. Many pharmaceutical and medtech companies have taken steps to improve visibility into supplier quality risks by using smart quality principles. For example, a large pharmaceutical company that implemented this data-driven approach eliminated in less than two years major CMO and supplier findings that were identified during audits.
AN ANALYSIS OF A SOFTWARE QUALITY ASSURANCE TOOL'S IMPLEMENTATION: A CASE STUDY. John J. Scarpino, Robert Morris University, scarpino@rmu Paul J. Kovacs, Robert Morris University, kovacs@rmu. ABSTRACT. Software Quality Assurance has grown within the last 10 years and now incorporates more sophisticated tools that enhance software testing ...